1
answer
0
watching
209
views
11 Dec 2019
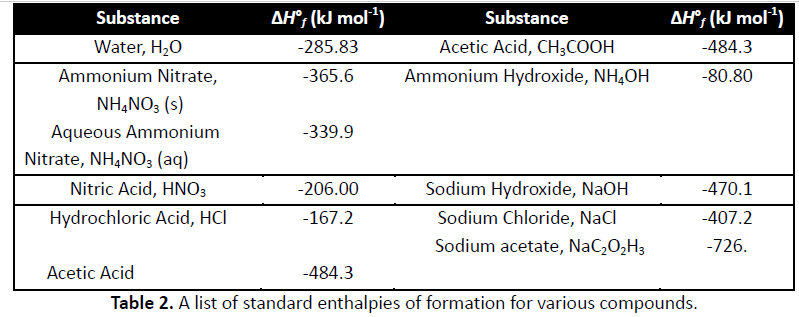
A solution of sodium acetate in water is observed to become more alkaline as the temperature is raised. Which conclusion can be drawn?. This is the equation for the reaction.

A. The forward reaction proceeds with the evolution of heat
B. The forward reaction proceeds with the absorption of heat
C. Acetic acid is less soluble in hot water than in cold water
D. At higher temperatures, 
Please explain your answer?
A solution of sodium acetate in water is observed to become more alkaline as the temperature is raised. Which conclusion can be drawn?. This is the equation for the reaction.

A. The forward reaction proceeds with the evolution of heat
B. The forward reaction proceeds with the absorption of heat
C. Acetic acid is less soluble in hot water than in cold water
D. At higher temperatures, 
Please explain your answer?
1
answer
0
watching
209
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Kottherva SreevidyaLv10
17 Oct 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232