1
answer
0
watching
142
views
28 Nov 2019
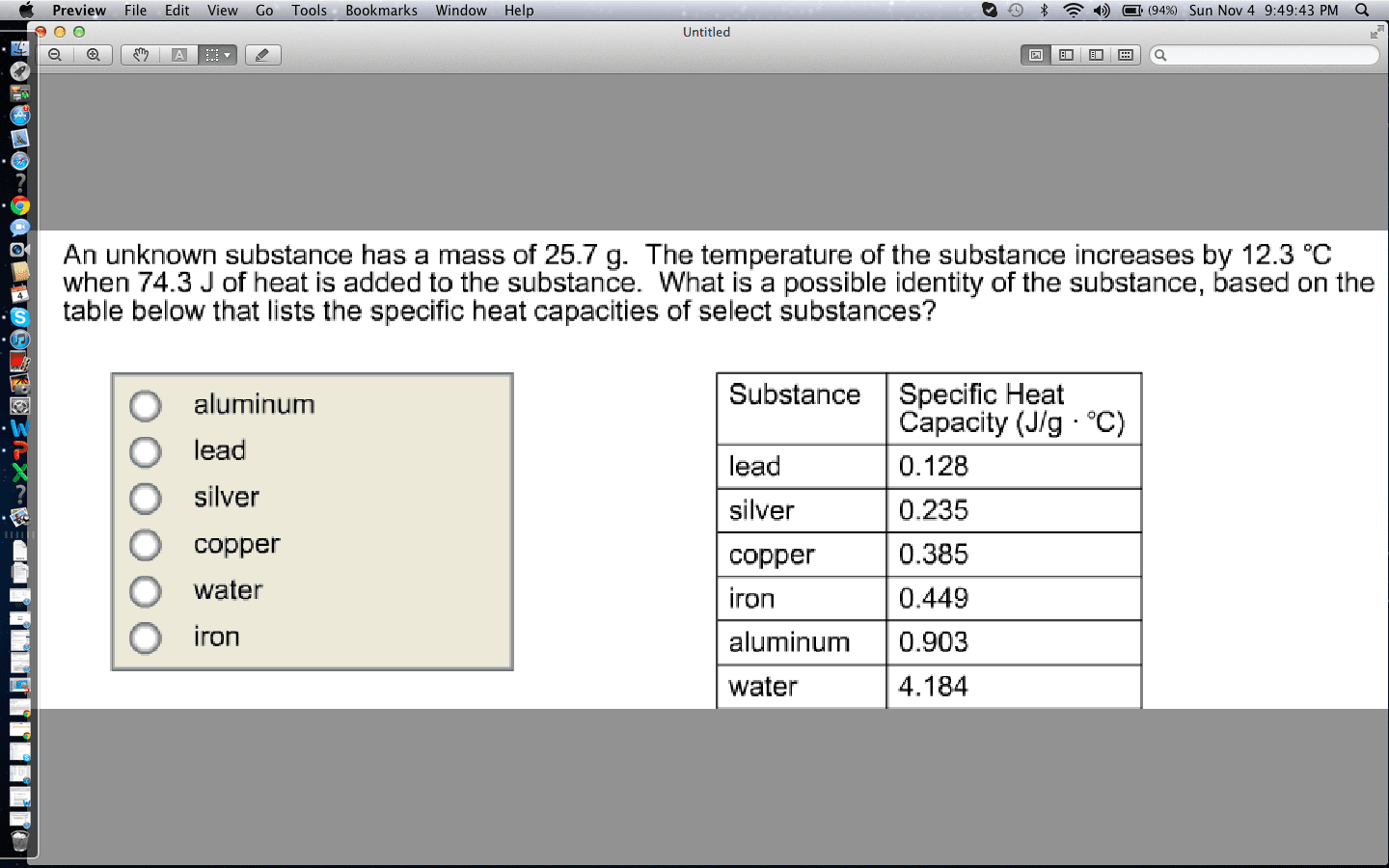
An unknown substance has a mass of 19.3 g. The temperature of the substance increases by 12.9 degrees Celsius when 58.5 J of heat is added to the substance. What is the most likely identity of the substance? The table lists the specific heats of select substances.
Substance... Specific Heat (J/g degrees Celsius)
lead.... 0.128
silver.... 0.235
copper.... 0.385
iron.... 0.449
aluminum.... 0.903
water.... 4.184
An unknown substance has a mass of 19.3 g. The temperature of the substance increases by 12.9 degrees Celsius when 58.5 J of heat is added to the substance. What is the most likely identity of the substance? The table lists the specific heats of select substances.
Substance... Specific Heat (J/g degrees Celsius)
lead.... 0.128
silver.... 0.235
copper.... 0.385
iron.... 0.449
aluminum.... 0.903
water.... 4.184
Keith LeannonLv2
12 Oct 2019