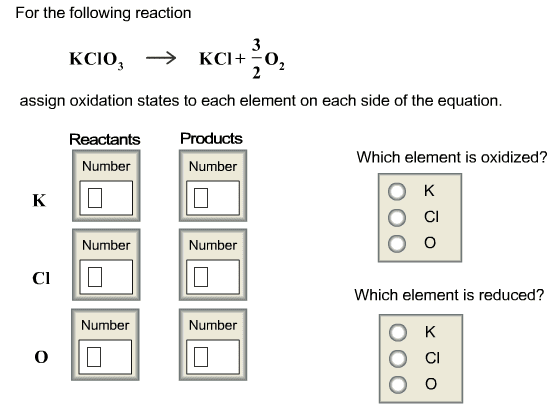
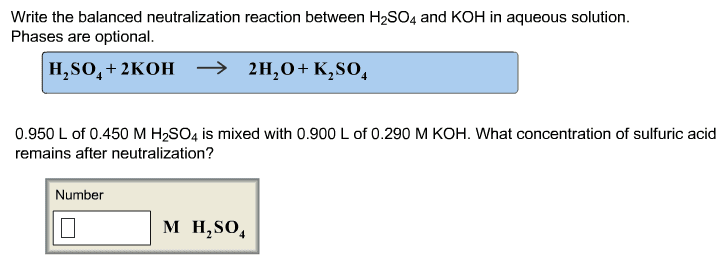
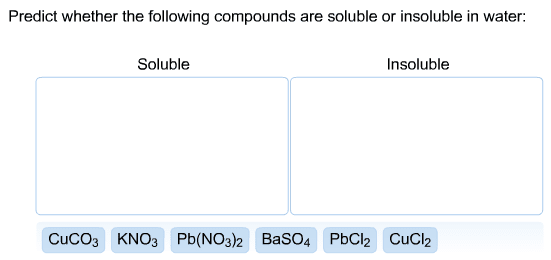
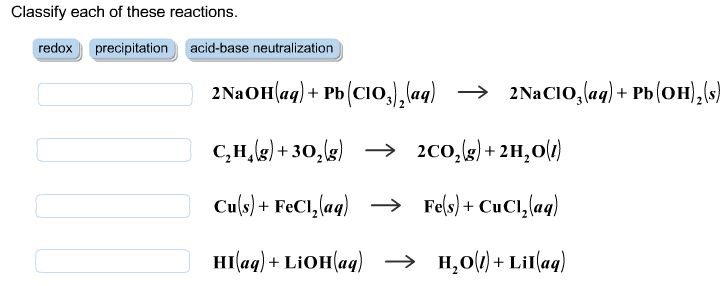1
answer
0
watching
468
views
26 Nov 2019
Oxalic acid, H2C2O4, is a toxicsubstnace found in spinach leaves. What is the molarity of asolution made by dissolving 12.0g of oxalic acid in enoguh water togive 400.0mL of solution? How many millilters of 0.100M KOH wouldyou need to titrate 25.0mL of the oxalic acid solution according tothe following equation?
H2C2O4(aq) +2KOH(aq) -->K2C2O4(aq) +2H2O(l)
Oxalic acid, H2C2O4, is a toxicsubstnace found in spinach leaves. What is the molarity of asolution made by dissolving 12.0g of oxalic acid in enoguh water togive 400.0mL of solution? How many millilters of 0.100M KOH wouldyou need to titrate 25.0mL of the oxalic acid solution according tothe following equation?
H2C2O4(aq) +2KOH(aq) -->K2C2O4(aq) +2H2O(l)
1
answer
0
watching
468
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Deanna HettingerLv2
12 Nov 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232