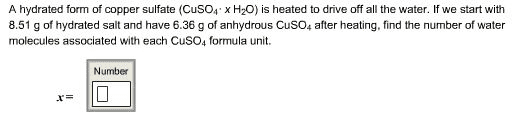1.A hydrated form of copper sulfate (CuSO4·x H2O) is heated to drive off all the water. If we start with 9.19 g of hydrated salt and have 5.87 g of anhydrous CuSO4 after heating, find the number of water molecules associated with each CuSO4 formula unit.
2.Iron(II) Sulfate forms a blue-green hydrate with the formula FeSO4 ·n H2O(s). If this hydrate is heated to a high enough temperature, H2O(g) can be driven off, leaving the dirty yellow anhydrous salt FeSO4(s). A 18.300-g sample of the hydrate was heated to 300 °C. The resulting FeSO4(s) had a mass of 9.9993 g. Calculate the value of n in FeSO4 ·n H2O(s).
3.The combustion of ethane (C2H6) produces carbon dioxide and steam:
2C2H6 (G)+7O2(G)------->4CO2 (G)+6H2O (G)
How many moles of CO2 are produced when 5.15 mol of ethane are burned in an excess of oxygen?
--------MOLS?
1.A hydrated form of copper sulfate (CuSO4·x H2O) is heated to drive off all the water. If we start with 9.19 g of hydrated salt and have 5.87 g of anhydrous CuSO4 after heating, find the number of water molecules associated with each CuSO4 formula unit.
2.Iron(II) Sulfate forms a blue-green hydrate with the formula FeSO4 ·n H2O(s). If this hydrate is heated to a high enough temperature, H2O(g) can be driven off, leaving the dirty yellow anhydrous salt FeSO4(s). A 18.300-g sample of the hydrate was heated to 300 °C. The resulting FeSO4(s) had a mass of 9.9993 g. Calculate the value of n in FeSO4 ·n H2O(s).
3.The combustion of ethane (C2H6) produces carbon dioxide and steam:
2C2H6 (G)+7O2(G)------->4CO2 (G)+6H2O (G)
How many moles of CO2 are produced when 5.15 mol of ethane are burned in an excess of oxygen?
--------MOLS?