1
answer
0
watching
152
views
23 Nov 2019
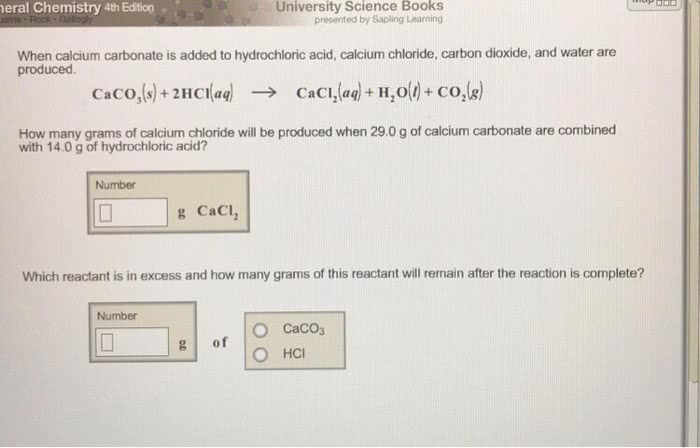
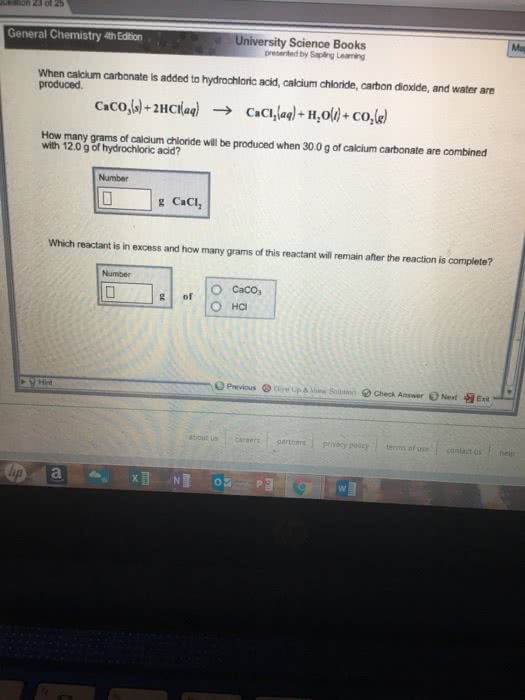
When calcium carbonate is added to HCl, calcium chloride, carbondioxide, & water are produced..
CaCO3 + 2HCl --> CaCl2 + H2O + CO2
How many grams of calcium chloride will be produced when 29.0 gramsof calcium carbonate are combined with 15.0 grams of HCl?
Which reactant is in excess & how many grams of this reactantwill remain after the reaction is complete?
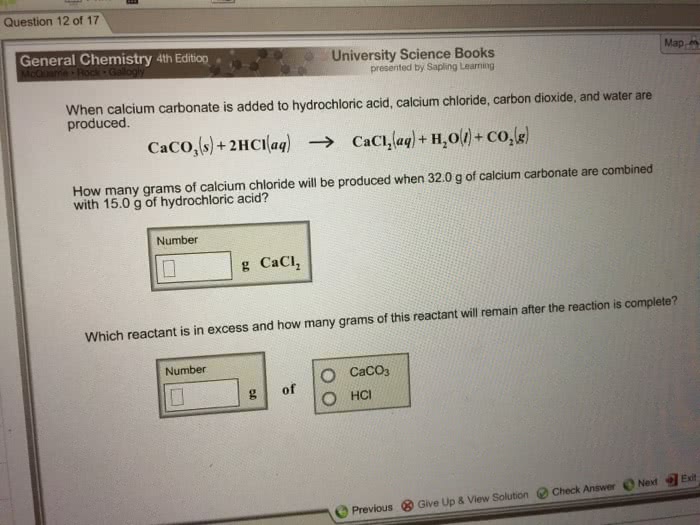
When calcium carbonate is added to HCl, calcium chloride, carbondioxide, & water are produced..
CaCO3 + 2HCl --> CaCl2 + H2O + CO2
How many grams of calcium chloride will be produced when 29.0 gramsof calcium carbonate are combined with 15.0 grams of HCl?
Which reactant is in excess & how many grams of this reactantwill remain after the reaction is complete?
Tod ThielLv2
21 Nov 2019