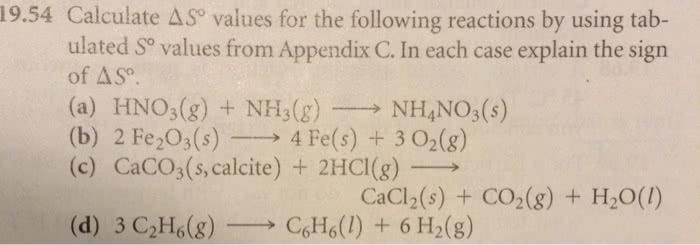1
answer
0
watching
103
views
23 Nov 2019
Using standard enthalpies of formation from Appendix L,calculate ?Ho for the following reaction: 2 C6H6(l) + 15 O2(g) ? 12CO2(g) + 6 H2O(g) b. How many grams of C6H6(l) must be combusted inthe above reaction to produce 2570 kJ of heat energy?
C6H6(l)= 48.95
o2 = O
CO2 = -393.509
H2O = -241.83
Using standard enthalpies of formation from Appendix L,calculate ?Ho for the following reaction: 2 C6H6(l) + 15 O2(g) ? 12CO2(g) + 6 H2O(g) b. How many grams of C6H6(l) must be combusted inthe above reaction to produce 2570 kJ of heat energy?
C6H6(l)= 48.95
o2 = O
CO2 = -393.509
H2O = -241.83
Sixta KovacekLv2
16 Nov 2019