1
answer
0
watching
187
views
23 Nov 2019
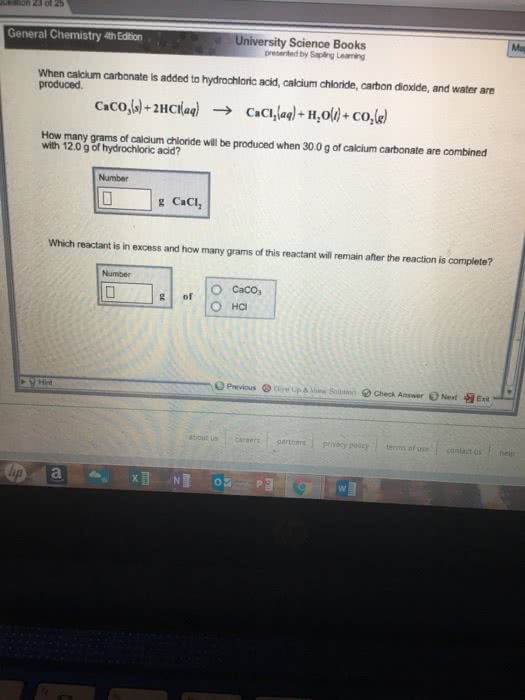
45g of Calcium carbonate is added to a 1.5L solution of hydrochloric acid with a density of 1.13 gHCl/mL. Assuming volume remains constant, what will the molar concentration of HCl be after the calcium carbonate is added?
45g of Calcium carbonate is added to a 1.5L solution of hydrochloric acid with a density of 1.13 gHCl/mL. Assuming volume remains constant, what will the molar concentration of HCl be after the calcium carbonate is added?
Bunny GreenfelderLv2
27 Apr 2019