1
answer
0
watching
538
views
23 Nov 2019
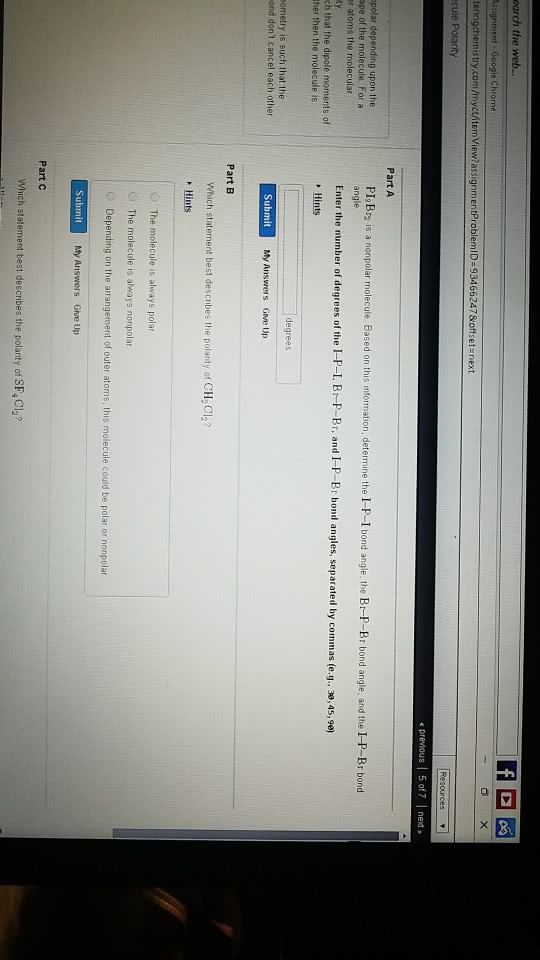
1. Bond polarity and molecular shape determine molecular polarity, which is measured as a dipole moment. True or False?
2. Molecular shape and polarity can affect physical properties, such as boiling point, and they play a central role in biological function. True or False?
3. A molecule with polar bonds is not necessarily a polar molecule. When bond polarities cancel each other, the molecule is nonpolar; when they reinforce each other, the molecule is polar. True or False?
1. Bond polarity and molecular shape determine molecular polarity, which is measured as a dipole moment. True or False?
2. Molecular shape and polarity can affect physical properties, such as boiling point, and they play a central role in biological function. True or False?
3. A molecule with polar bonds is not necessarily a polar molecule. When bond polarities cancel each other, the molecule is nonpolar; when they reinforce each other, the molecule is polar. True or False?
Beverley SmithLv2
1 Jul 2019