1
answer
0
watching
423
views
20 Nov 2019
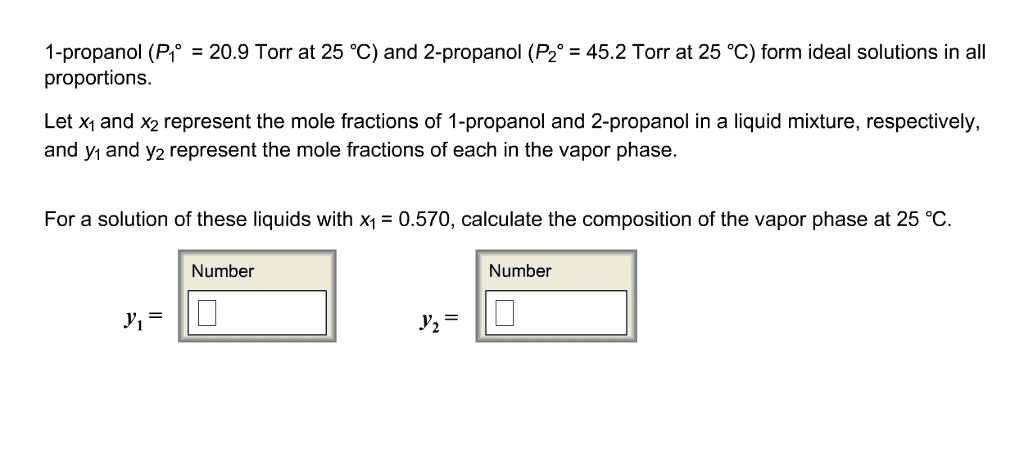
The vapor pressure of pure toluene is 400 torr and that of pure1 ,2-dimethyl benzeneis 150 torr at 90 degree C.Determine thecomposition (mole fractions) of the liquid and vapor phases iftoluene and 1, 2- dimethylbenzene are mixed at 90 degree C and atotal pressure of .5 atm .Assume ideal solution behavior. (rememberthe mole fraction in liquid and in vapor phase will be same)
The vapor pressure of pure toluene is 400 torr and that of pure1 ,2-dimethyl benzeneis 150 torr at 90 degree C.Determine thecomposition (mole fractions) of the liquid and vapor phases iftoluene and 1, 2- dimethylbenzene are mixed at 90 degree C and atotal pressure of .5 atm .Assume ideal solution behavior. (rememberthe mole fraction in liquid and in vapor phase will be same)
Beverley SmithLv2
7 Oct 2019