1
answer
0
watching
109
views
20 Nov 2019
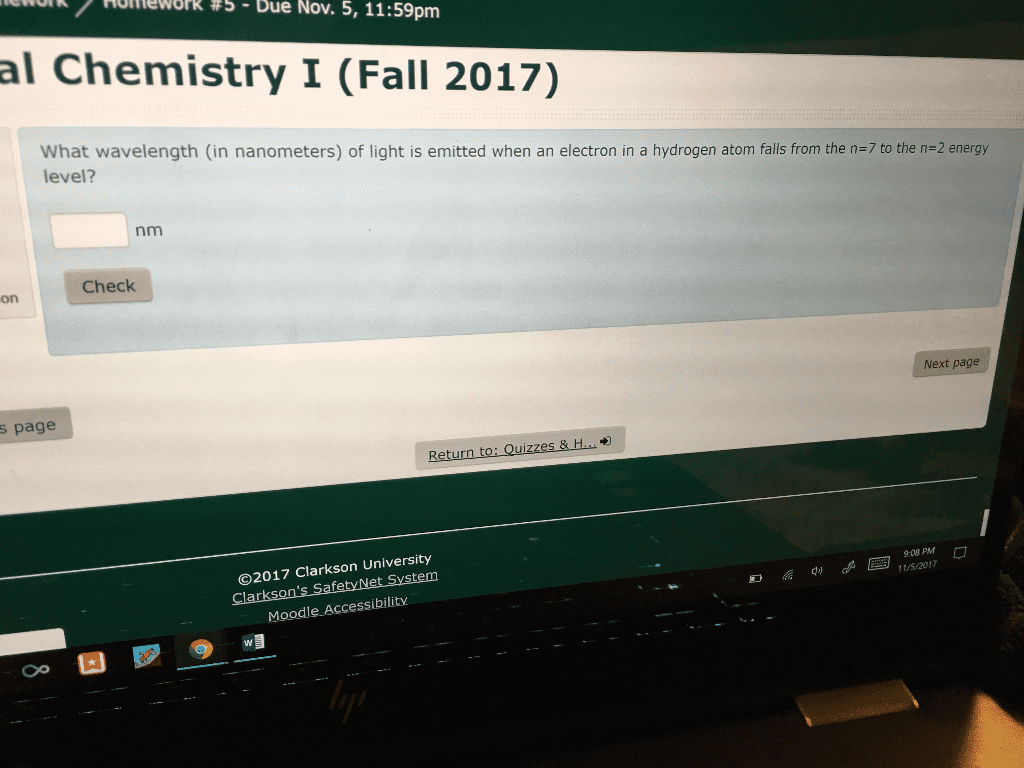
Calculate the energy and the wavelength, in nanometers, of light emitted when an electron in an excited hydrogen atom falls from the n=2 level to the n = 1 level.Can someone detail how you would figure out this problem?
Calculate the energy and the wavelength, in nanometers, of light emitted when an electron in an excited hydrogen atom falls from the n=2 level to the n = 1 level.Can someone detail how you would figure out this problem?
Keith LeannonLv2
8 Apr 2019