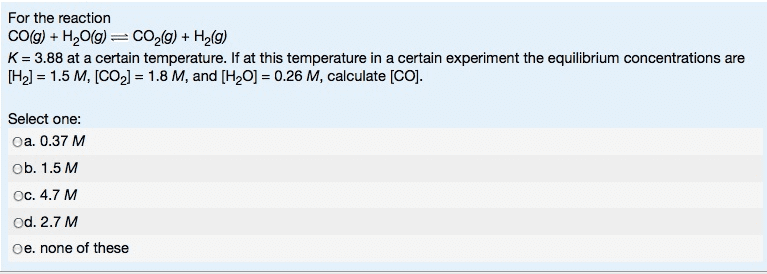
1
answer
0
watching
603
views
19 Nov 2019
CaO(s)+CO2(g)=CaCO3(s)
given the equilibrium constant Kc is .6*10^2 ,caculate the mass of CaCO3 when 3.00 g of CaO AND 2.50g of CO2 are combined in 2.50L closed rigid container
what is the mass of CaCO3 at equilibrium
CaO(s)+CO2(g)=CaCO3(s)
given the equilibrium constant Kc is .6*10^2 ,caculate the mass of CaCO3 when 3.00 g of CaO AND 2.50g of CO2 are combined in 2.50L closed rigid container
what is the mass of CaCO3 at equilibrium
Patrina SchowalterLv2
9 Apr 2019