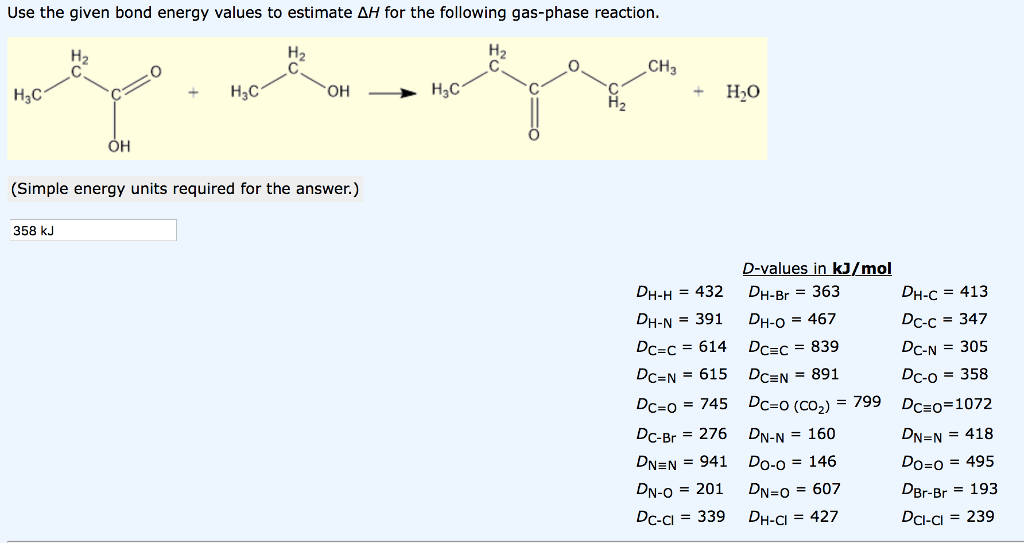
1
answer
0
watching
373
views
19 Nov 2019
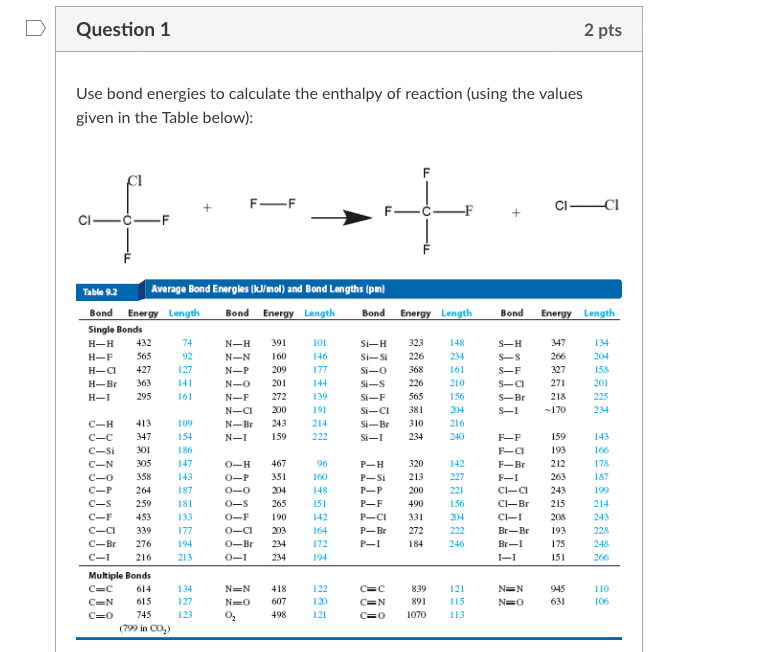
Question 1 Use bond energies to calculate the enthalpy of reaction (using the values given in the Table below): +CICi Table 9.2 Average Bond Energles (kmol) and Bond Lengths (pm Bond Energy Leng Single Bonds H-H 432 H-F 565 H-C 427 H-Br 363 H-I Bond N-H 391 N-N 160 N-P 209 N-O 201 N-F 27239 N-CI200 N-Br 243 N-I 59222 Si-H 323 Si- Si 226 Si-o 368 S-S 226 Si-F 565 Si-C 381 Si-Br310 S-H S-S S-F S-C271 S-Br 218 S-I 146 161 327 158 144 201 295 161 156 191 234 109 154 C-H 413 C-c 347 C-si 301 C-N 305 C-O 358 C-P 264 C-s 259 C-F 453 C-Ci 339 C-Br 276 194 C-I Multiple Bonds C-c 614 C-N 615 C=0 745 240 F-F 143 0-H 467 o-P 331 0-0 204 o-s 265 O-F 190 147 143 178 187 96 P-H P-Si 213 P-P P-F P-CI 331 P-Br272 F-Br 212 F-I C-CI243 C-Br 215 C-I Br-Br Br-I I-I 156 181 133 243 193 172 248 O-Br 234 0- 234 122 120 121 N=N 418 839 12 891 115 134 N-N 607 498 N=O 631 106 C O 1070 113 (799 in CO

Question 1 Use bond energies to calculate the enthalpy of reaction (using the values given in the Table below): +CICi Table 9.2 Average Bond Energles (kmol) and Bond Lengths (pm Bond Energy Leng Single Bonds H-H 432 H-F 565 H-C 427 H-Br 363 H-I Bond N-H 391 N-N 160 N-P 209 N-O 201 N-F 27239 N-CI200 N-Br 243 N-I 59222 Si-H 323 Si- Si 226 Si-o 368 S-S 226 Si-F 565 Si-C 381 Si-Br310 S-H S-S S-F S-C271 S-Br 218 S-I 146 161 327 158 144 201 295 161 156 191 234 109 154 C-H 413 C-c 347 C-si 301 C-N 305 C-O 358 C-P 264 C-s 259 C-F 453 C-Ci 339 C-Br 276 194 C-I Multiple Bonds C-c 614 C-N 615 C=0 745 240 F-F 143 0-H 467 o-P 331 0-0 204 o-s 265 O-F 190 147 143 178 187 96 P-H P-Si 213 P-P P-F P-CI 331 P-Br272 F-Br 212 F-I C-CI243 C-Br 215 C-I Br-Br Br-I I-I 156 181 133 243 193 172 248 O-Br 234 0- 234 122 120 121 N=N 418 839 12 891 115 134 N-N 607 498 N=O 631 106 C O 1070 113 (799 in CO
Bunny GreenfelderLv2
24 Feb 2019