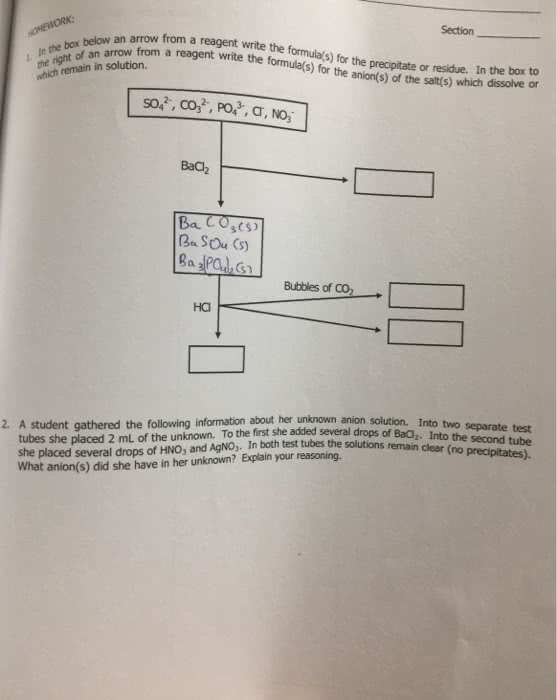
Anyone have any idea how to answer this..?


For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
URGENT:
Attached please see the results and the data tables needed.
1) Use your results and observations in Data Table 1 to create a flow chart for identification of unknown anions, using the AgNO3 and HCl confirmation tests. Insert the flow chart below.
2)Use your results and observations in Data Tables 1, 2, and 3, to create a flow chart for the identification of an unknown (both an unknown anion and cation), using the AgNO3 and HCl confirmation tests and the cation flame tests.
Thank you
Data Table 1. Anion Confirmation Tests.
| Chemical | Anion | Addition of AgNO3 | Addition of HCl |
| NaBr | Bromide | The solution became white and cloudy. | The solution remained white with its cloudy appearance. |
| Na2CO3 | Carbonate | The inside is clear while the outside is cloudy. | The solution remained white and cloudy. Bubbles formed. |
| NaCl | Chloride | The bottom of the solution turned a little cloudy while the top remained clear. | The solution remained white and cloudy on the bottom and clear on the top. |
| NaI | Iodide | In the middle it is dark and cloudy. It fades out and around the middle is a lighter white. | The solution became a little separated. |
| Na2SO4 | Sulfate | The solution is perfectly clear. | The solution turned into a white color and a little cloudy appearance. |
| Na2S | Sulfide | It turns into a brownish color in the middle but separated from the clear. | The brown continues to become more separated from the clear. |
| Unknown #103 | carbonate | White and a little cloudy. It contains small particles. | White and cloudy. Bubbles formed throughout the solution. |
| Unknown #104 | sulfide | Brown color in the middle. The solution around it remained clear. | Some of the brown coloring disappeared and become separate. |
Data Table 2. Cation Flame Tests.
| Chemical | Cation | Flame Observations |
| KI | Potassium | Light pink |
| CaCl2 | Calcium | Red |
| LiOH | Lithium | Fuchsia pink |
| NaCl | Sodium | Orange |
| Cu(NO3)2 | Copper | Turquoise |
| Unknown #105 | Copper | Turquoise |
| Unknown #106 | Potassium | Light pink |
| Unknown #107 | Calcium | Red |
Data Table 3. Complete Chemical Identification.
| Chemical | Cation | Anion Test Observations | Anion | Complete Chemical Name and Formula |
| Unknown #105 | Copper | With AgNO3 there is no change that can be observed. | CO32- | Copper(II) nitrate, Cu(NO3)2 |
| Unknown #106 | Potassium | With AgNO3 the middle is a very dark color while the outside is white and cloudy. | I- | Potassium iodide, KI |
| Unknown #107 | Calcium | With AgNO3 it turned into a white and cloudy solution. | Cl- | Calcium chloride,CaCl2 |