1
answer
0
watching
655
views
18 Nov 2019
Please answer step by step and explain.
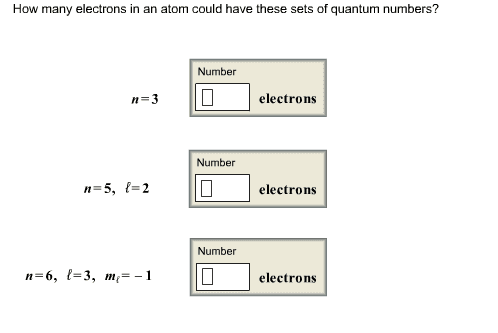
How many electrons in an atom could have these sets of quantum numbers? n = 3 n = 5, l = 2 n = 6, l = 3, m_l = -1
Please answer step by step and explain.

How many electrons in an atom could have these sets of quantum numbers? n = 3 n = 5, l = 2 n = 6, l = 3, m_l = -1
Elin HesselLv2
14 Jan 2019
