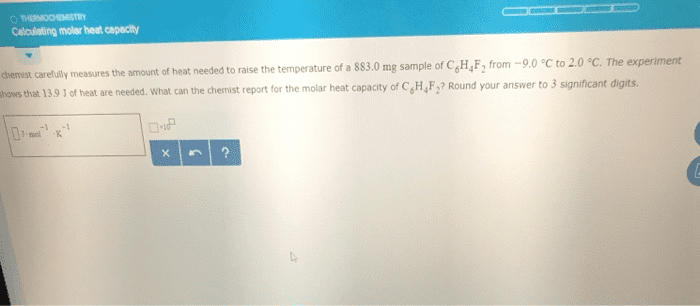
1
answer
0
watching
486
views
18 Nov 2019
A chemist carefully measures the amount of heat needed to raise the temperature of a 0.27kg sample of a pure substance from â2.5°C to 12.7°C . The experiment shows that 0.96kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Be sure your answer has the correct number of significant digits.
A chemist carefully measures the amount of heat needed to raise the temperature of a 0.27kg sample of a pure substance from â2.5°C to 12.7°C . The experiment shows that 0.96kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Be sure your answer has the correct number of significant digits.
Bunny GreenfelderLv2
11 Jul 2019