1
answer
0
watching
212
views
18 Nov 2019
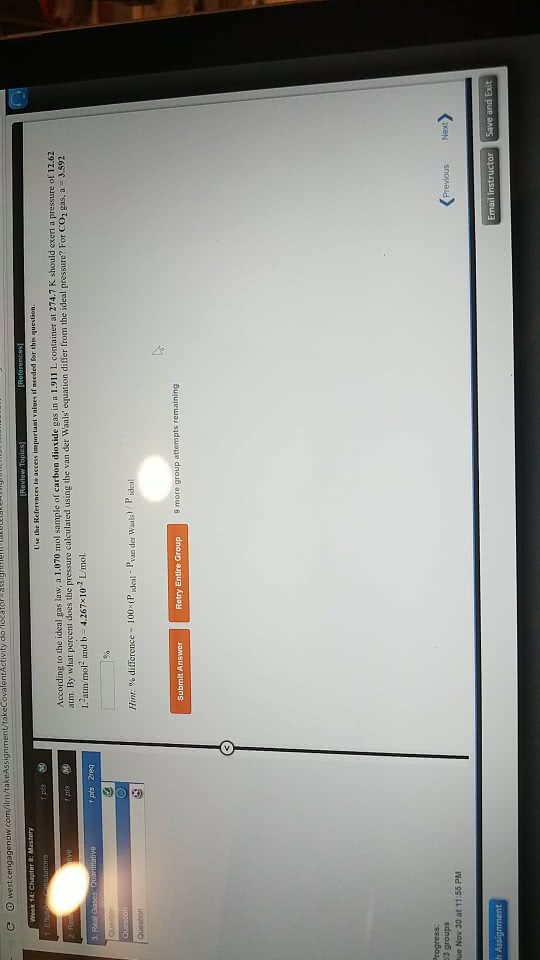
According to the ideal gas law, a 10.61 mol sample of oxygen gas in a 0.8199 L container at 504.7 K should exert a pressure of 535.9 atm. By what percent does the pressure calculated using the van der Waals' equation differ from the ideal pressure? For O2 gas, a = 1.360 L2atm/mol2 and b = 3.183Ã10-2 L/mol.
According to the ideal gas law, a 10.61 mol sample of oxygen gas in a 0.8199 L container at 504.7 K should exert a pressure of 535.9 atm. By what percent does the pressure calculated using the van der Waals' equation differ from the ideal pressure? For O2 gas, a = 1.360 L2atm/mol2 and b = 3.183Ã10-2 L/mol.
Tod ThielLv2
6 Jun 2019