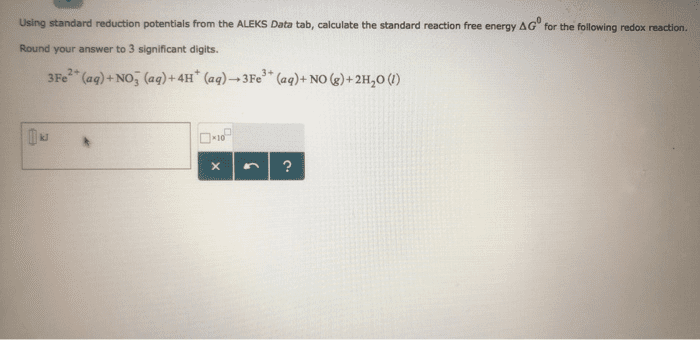
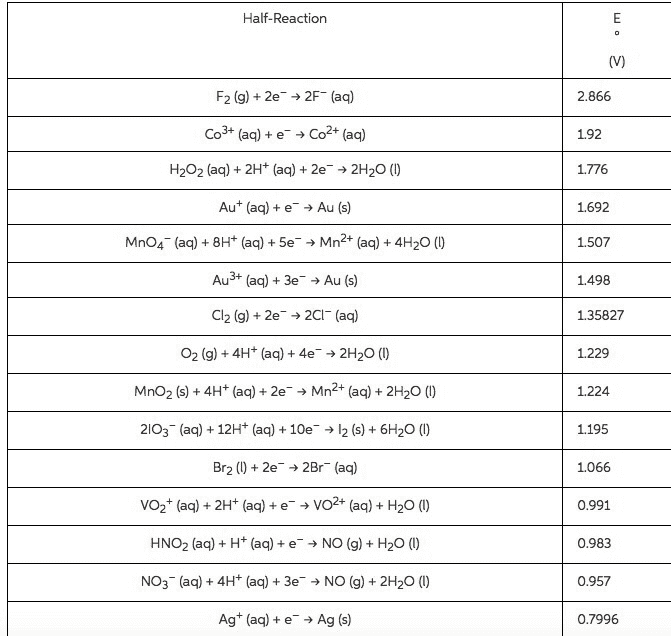
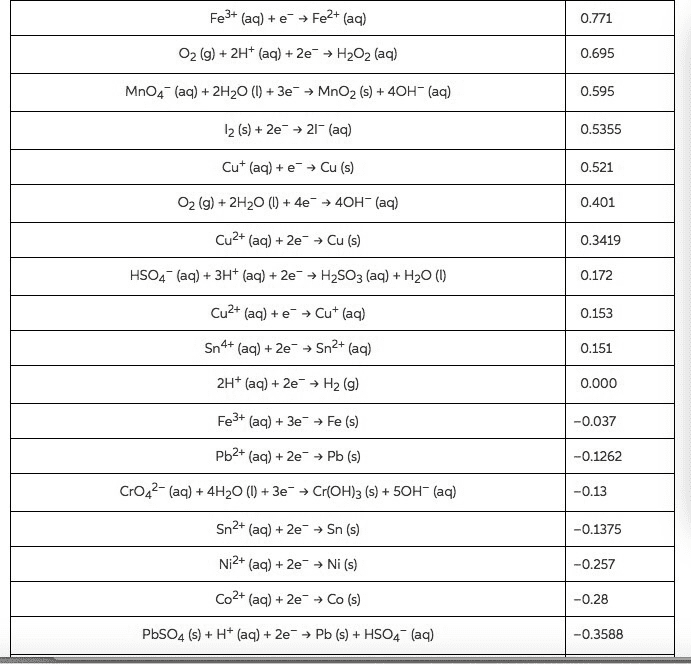
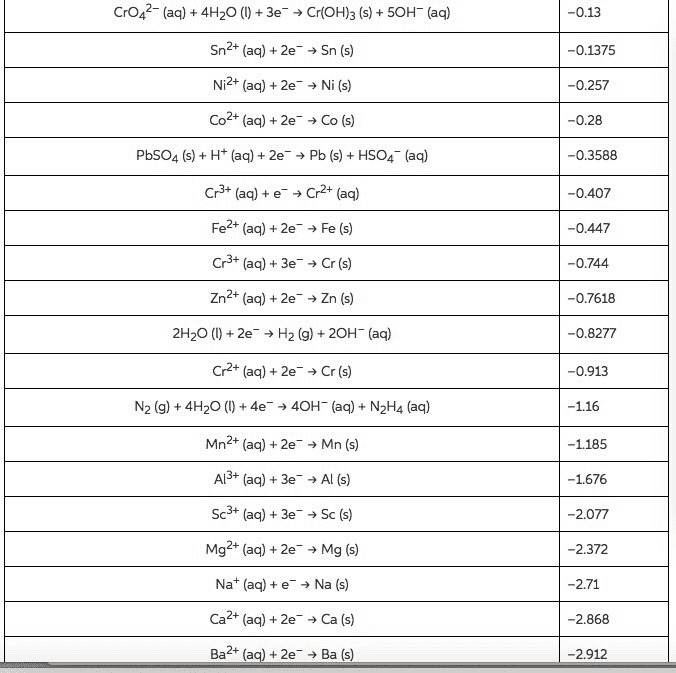
1
answer
0
watching
2,001
views
18 Nov 2019
What is the standard emf of a galvanic cell made of a Co electrode in a 1.0 M Co(NO3)2 solution and a Al electrode in a 1.0 M Al(NO3)3 
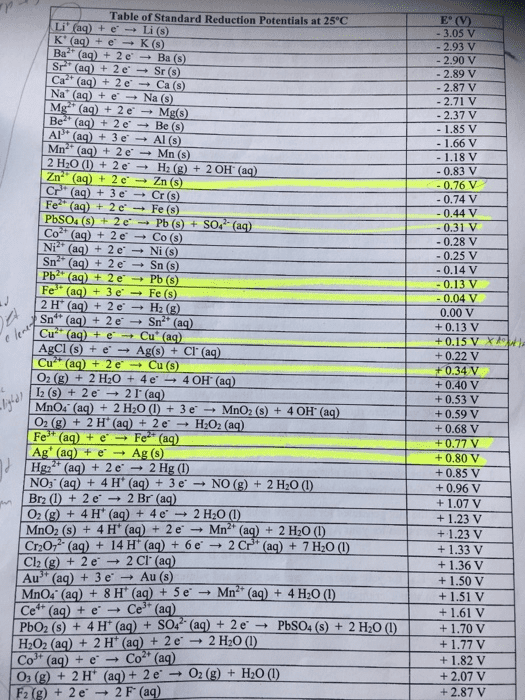
Standard Reduction Potentials at 25°C E(V) 2.87 +2.07 +1.82 Half-Reaction 03(g) + 2H+(aq) + 2e-_âO2(g) + H2O Co3+(aq) + e-â Co2+(aq) o (a H2O2(aq) + 2H+(aq) 2e--â 2H20 Cu2+(aq) + 2e- Cu(s) AgCl(s) + e--Ag(s) + Cl"(aq) SO'"(aq) + 4H'(aq) + 2e-_â SOgg) + 2H2O Cu2+(aq) + e-_ Cu+(aq) sn'+(aq) + 2e-_â Sn2+(aq) 2H+(aq) + 2e H2(g) Pb2+(aq) + 2e"-â Pb(s) Sn2(aq) 2e Sn(s) Ni2+(aq) + 2e Ni(s) +0.34 0.22 +0.20 +0.15 å0.13 0.00 0.13 0.14 0.25 -0.28 0.31 0.40 0.44 0.74 0.76 0.83 1.18 1.66 1.85 Cd2+(aq) + 2e-â Cd(5) Fe2+(aq) + 2e â Fe(s) â Cr(s) Zn2+(aq) + 2e-_ â Zn(s) 2H20 2eH2(g) +20H (aq) Mn2+(aq) + 2e â Mn(s) Al(a) 3eAI(s) 2+
What is the standard emf of a galvanic cell made of a Co electrode in a 1.0 M Co(NO3)2 solution and a Al electrode in a 1.0 M Al(NO3)3 
Standard Reduction Potentials at 25°C E(V) 2.87 +2.07 +1.82 Half-Reaction 03(g) + 2H+(aq) + 2e-_âO2(g) + H2O Co3+(aq) + e-â Co2+(aq) o (a H2O2(aq) + 2H+(aq) 2e--â 2H20 Cu2+(aq) + 2e- Cu(s) AgCl(s) + e--Ag(s) + Cl"(aq) SO'"(aq) + 4H'(aq) + 2e-_â SOgg) + 2H2O Cu2+(aq) + e-_ Cu+(aq) sn'+(aq) + 2e-_â Sn2+(aq) 2H+(aq) + 2e H2(g) Pb2+(aq) + 2e"-â Pb(s) Sn2(aq) 2e Sn(s) Ni2+(aq) + 2e Ni(s) +0.34 0.22 +0.20 +0.15 å0.13 0.00 0.13 0.14 0.25 -0.28 0.31 0.40 0.44 0.74 0.76 0.83 1.18 1.66 1.85 Cd2+(aq) + 2e-â Cd(5) Fe2+(aq) + 2e â Fe(s) â Cr(s) Zn2+(aq) + 2e-_ â Zn(s) 2H20 2eH2(g) +20H (aq) Mn2+(aq) + 2e â Mn(s) Al(a) 3eAI(s) 2+
1
answer
0
watching
2,001
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Casey DurganLv2
6 Jun 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232