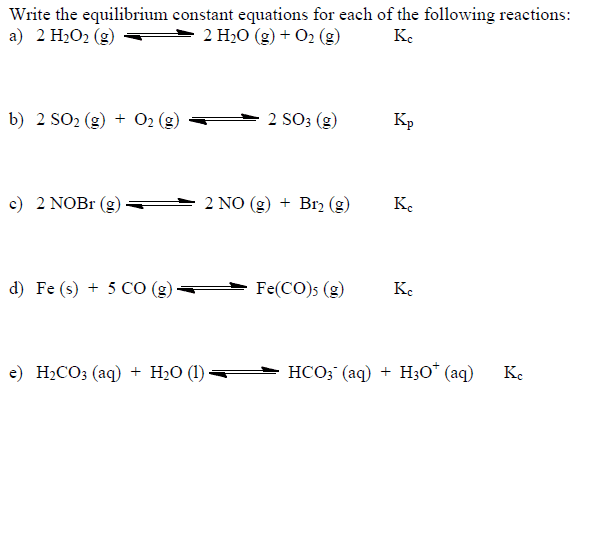1
answer
0
watching
153
views
18 Nov 2019


Write the equilibrium expression for K_c for the following reaction: N_2 (g) + 3H_2 (g) 2NH_3 (g) Remember to include the constant (K) in your expression K_c = [NH_3]^2/[N_2][H_2]^3 Write the equilibrium expressions (for K_c) for the following reactions. 1. 2NO_2 (g) N_2 O_2 (g) 2. H_2 (g) + I_2 (g) 2HI (g) 3. CaSO_4 (s) Ca^2+ (aq) + SO_4^3- (aq) 4. Mg(s) + 2Ag^+ (aq) Mg^2+ (aq) + 2Ag(s) 5. C(s) + H_2O (g) CO (g) + H_2 (g) 6. CO_2 (aq) + H_2 O (l) H^+ (aq) + HCO_3^+ (aq) 7. CO (g) + 2H_2 (g) CH_3 OH(g) 8. 2POCl_5 (g) 2PCl_3 (g) + O_2 (g) 9. Sn (s) + 2CO_2 (g) SnO_2 (g) + 2CO (g) 10. 2HBr (g) H_2 (g) + Br_2 (l) 11. O_2 (g) + NO_2 (g) O_3 (g) + NO (g) 12. Fe (s) + H_3O (g) FeO (s) + H_2 (g) Balance the following equations. Write the equilibrium expressions (for K_c) for each reaction. 1. _____ O_3 (g) _____ O_2 (g) 14. _____ NO (g) + _____ Cl_2 (g) _____ NOCl (g) 15. _____ CO (g) _____ C (s) + _____ O_2 (g) 16. _____ SO_3 (g) _____ SO_2 (g)
Collen VonLv2
3 Feb 2019