1
answer
0
watching
123
views
17 Nov 2019
I need help with 7and 8. Can you please show work

I need help with 7and 8. Can you please show work


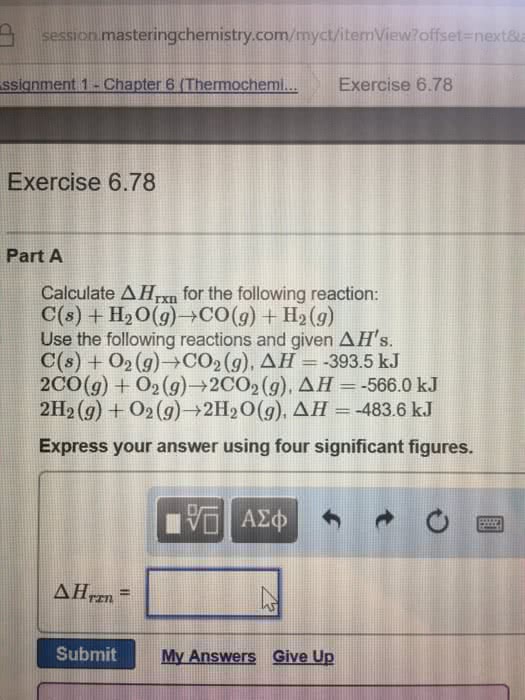
Using the following reactions and Delta H's: C_5H_12 (l) + 8O_2 (g) rightarrow 5CO_2 (g) + 6H_2O (l) Delta H = -3505.8 kJ C (s) + O_2 (g) rightarrow CO_2 (g) Delta H = -393.5 kJ 2H_2 (g) + O_2 (g) rightarrow 2H_2O (l) Delta H = -483.5 kJ Calculate Delta H_rxn for the following reaction: 5C (s) + 6H_2 (g) rightarrow C_5H_12 (l) Given: Delta Hf^o for [H_2O (l) = -286 kJ/mol; NO (g) = 90 kJ/mol; NO_2 (g) = 34 kJ/mol; HNO_3 = -207 kJ/mol Calculate Delta H_rxn^o for the reaction: 3NO_2 (g) + H_2O (l) rightarrow 2HNO_3 (aq) + NO (g)
Jean KeelingLv2
20 Sep 2019