



For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
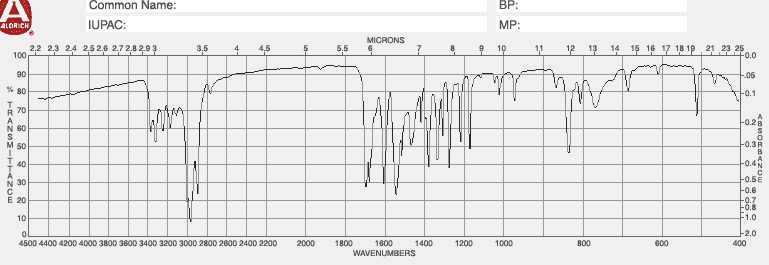
| IR List position (cm-1) & functional group | 4. |
| 1. | 5. |
| 2. | 6. |
| 3. | 7. |
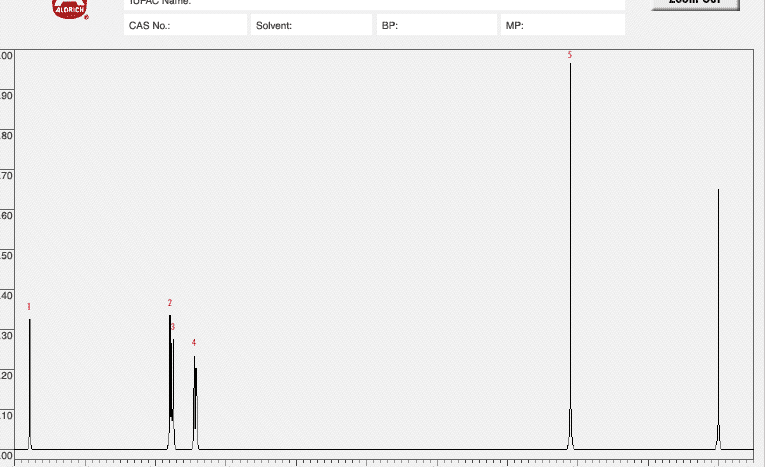
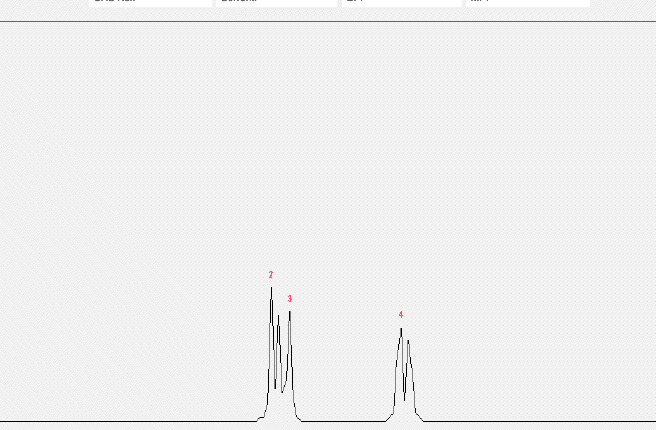
| Peak | Chemical Shift (δ) | Multiplicityâ | Hâ¡ | Peak | Chemical Shift (δ) | Multiplicityâ | Hâ¡ |
| 1. | 7. | ||||||
| 2. | 1 | 8. | |||||
| 3. | 1 | 9. | |||||
| 4. | 6 | 10. | |||||
| 5. | 11. | ||||||
| 6. | 12. |
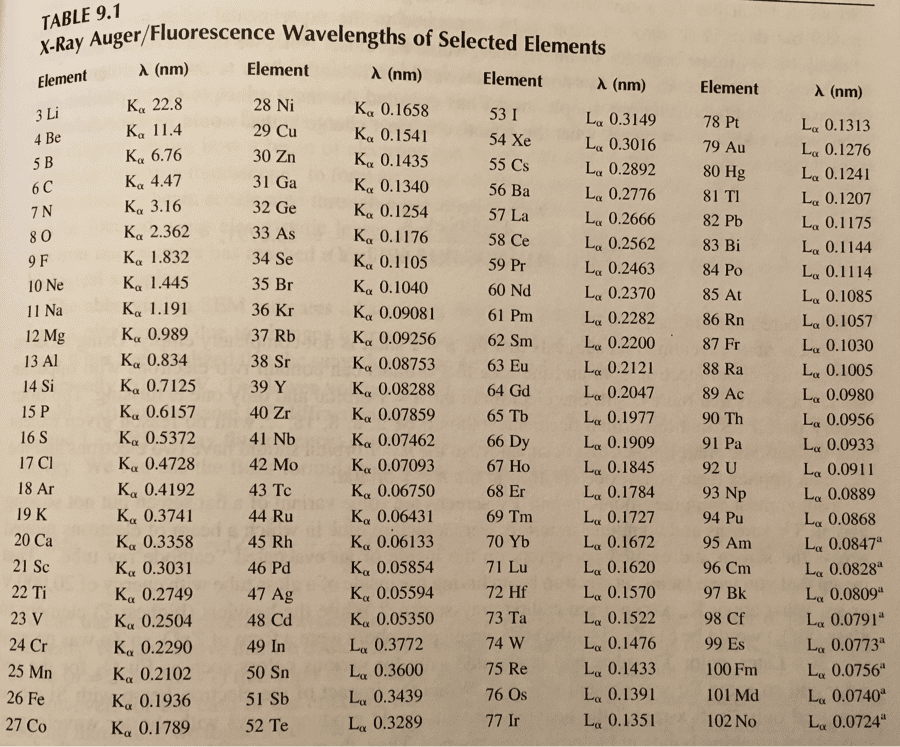
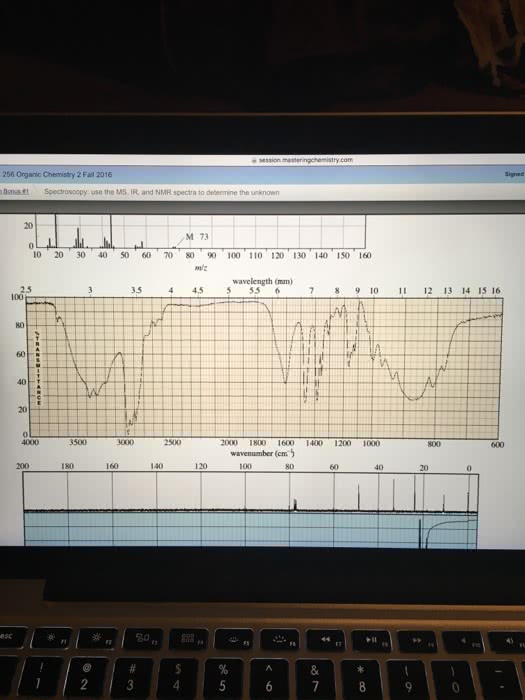
Please fill out the NMR and IR tables for each spectroscopy.
BP 300,dec
%C 66.25
%H 5.56
Melting point 157 degrees
can you determine the unknown?
There are 25 peaks (28 if you count the solvent) in the 13C NMR spectrum of trifluoroacetylguaiazulene, even though one might expect 16 peaks based on the number of chemically distinct carbons. What accounts for the extra peaks? (and just in case youâre wondering: NO, the extra peaks are not due to impurities) List the chemical shift of all 16 carbons. Note: Peak at 77 is due to CDCl3, peak at 135 is a doublet peak, and peak at 141 is a doublet of doublet.
13C NMR spectrum (150 MHz, CDCl3) of trifluoroacetylguaiazulene:
Peak listing of the 13C NMR spectrum
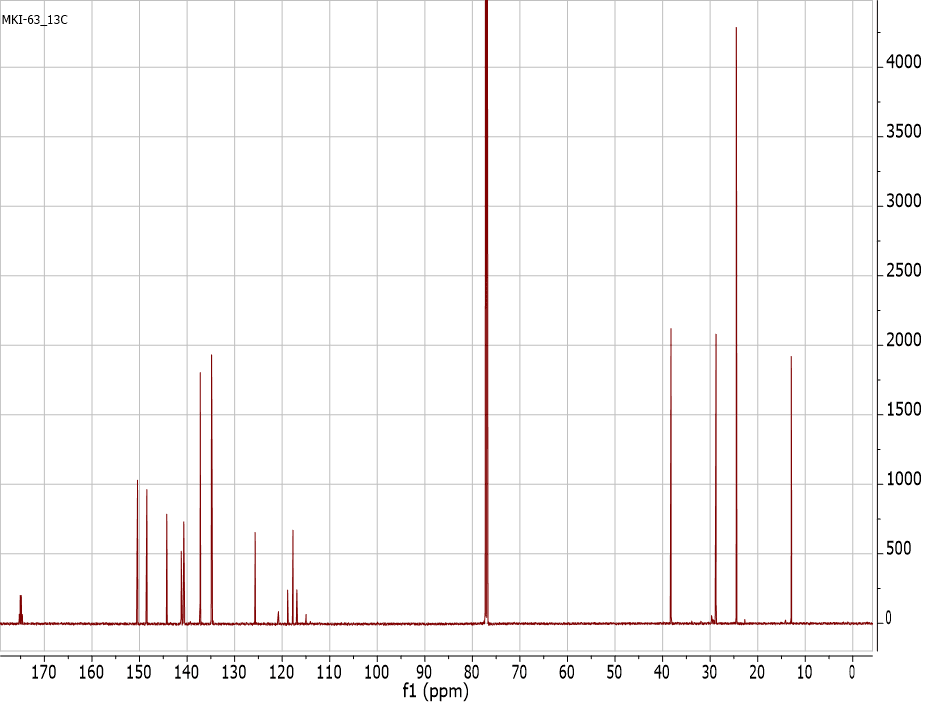
of trifluoroacetylguaiazulene.
| Peak | ppm | Hz |
| 1 | 175.30 | 26451.1 |
| 2 | 175.08 | 26418.2 |
| 3 | 174.86 | 26385.1 |
| 4 | 174.65 | 26352.1 |
| 5 | 150.42 | 22696.7 |
| 6 | 148.46 | 22400.6 |
| 7 | 144.27 | 21768.6 |
| 8 | 141.22 | 21308.9 |
| 9 | 140.74 | 21235.3 |
| 10 | 140.71 | 21231.7 |
| 11 | 140.69 | 21228.1 |
| 12 | 140.66 | 21224.4 |
| 13 | 137.23 | 20706.9 |
| 14 | 134.84 | 20345.2 |
| 15 | 134.72 | 20328.3 |
| 16 | 125.68 | 18963.5 |
| 17 | 120.79 | 18225.9 |
| 18 | 118.85 | 17933.4 |
| 19 | 117.73 | 17764.3 |
| 20 | 116.91 | 17640.9 |
| 21 | 114.98 | 17348.4 |
| 22 | 77.25 | 11656.0 |
| 23 | 77.04 | 11624.1 |
| 24 | 76.83 | 11592.1 |
| 25 | 38.23 | 5767.8 |
| 26 | 28.75 | 4337.7 |
| 27 | 24.48 | 3693.6 |
| 28 | 12.94 | 1952.0 |