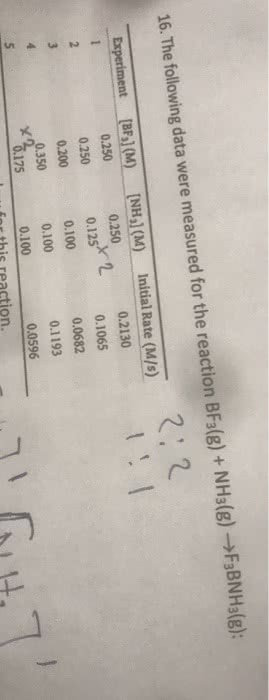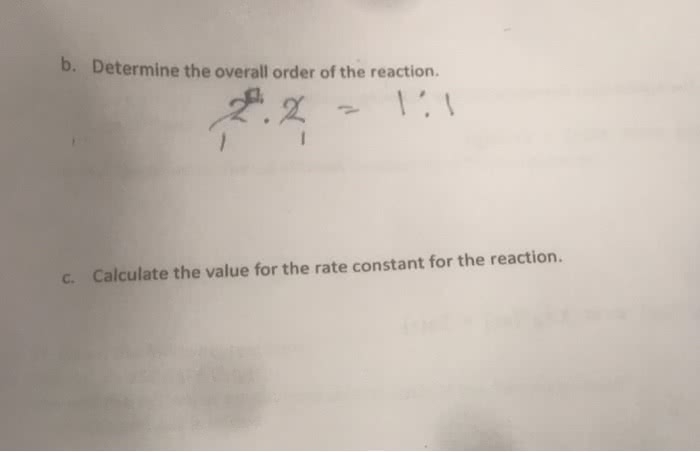2
answers
0
watching
203
views
28 Sep 2019
The data in the table below were collected for the gas phase reaction shown
BF3(g) + NH3(g) > F3BNH3(g)
Experiment [BF3] (M) [NH3] (M) Initial Rate (m/s)
1 0.500 0.250 0.4262
2 0.500 0.125 0.2131
3 0.400 0.200 0.2728
4 0.700 0.200 0.4772
5 0.350 0.200 0.2384
1) Calculate the reaction orders for each reactant. 2) State the overall reaction order. 3) Determine the rate law for this reaction (including k).
The data in the table below were collected for the gas phase reaction shown
BF3(g) + NH3(g) > F3BNH3(g)
Experiment [BF3] (M) [NH3] (M) Initial Rate (m/s)
1 0.500 0.250 0.4262
2 0.500 0.125 0.2131
3 0.400 0.200 0.2728
4 0.700 0.200 0.4772
5 0.350 0.200 0.2384
1) Calculate the reaction orders for each reactant. 2) State the overall reaction order. 3) Determine the rate law for this reaction (including k).
25 Apr 2023
Tod ThielLv2
28 Sep 2019
Already have an account? Log in