1
answer
0
watching
520
views
28 Sep 2019
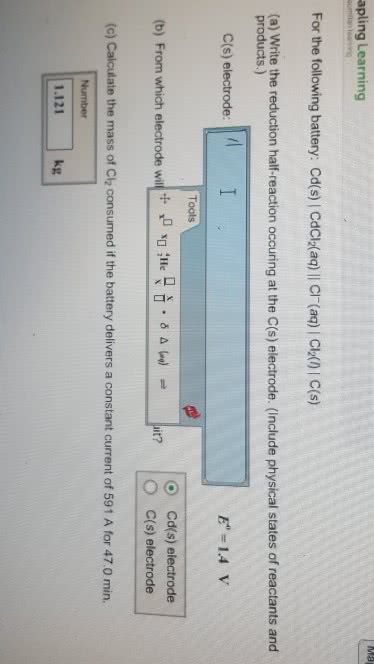
for the following battery: Cd(s)line CdCl2(aq)double lineCl^-(aq) line Cl2(l) line C(s)
A) Write the reduction half reaction occuring at the C(s)electrode . (Include physical states of reactants andproducts.)
C(s) electrode: please provide.
E^*=1.4 V
B) From which electrode will electrons flow from the batteryinto a circuit.
1. Cd(s) electrode
2.C(s) electrode
C) calculate the mass of Cl2 consumed if the battery delivers aconstant current of 713 A for 30.0 min.
Answer must be in Kg.
for the following battery: Cd(s)line CdCl2(aq)double lineCl^-(aq) line Cl2(l) line C(s)
A) Write the reduction half reaction occuring at the C(s)electrode . (Include physical states of reactants andproducts.)
C(s) electrode: please provide.
E^*=1.4 V
B) From which electrode will electrons flow from the batteryinto a circuit.
1. Cd(s) electrode
2.C(s) electrode
C) calculate the mass of Cl2 consumed if the battery delivers aconstant current of 713 A for 30.0 min.
Answer must be in Kg.
Irving HeathcoteLv2
28 Sep 2019