1
answer
0
watching
195
views
mauvewolf23Lv1
28 Sep 2019
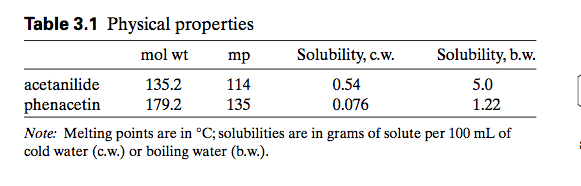
1. what is the minimum volume of boiling water needed todissolve .2 g of sodium benzoate? the olubility of sodium benzoatein boiling water is 1 gram per 1.4 mL. about how much sodiumbenzoate at 25 C is 1 gram per 1.8 mL. Calculate the max mass ofsolid sodium benzoate that can be recovered when the cooledsolution is filtered.
2. why would mixed solvents be used for recrystallization?
3. explain why the melting point of pure phthalic acid is higherthan that of pure naphthalene, when both have nearly the sameweight.
1. what is the minimum volume of boiling water needed todissolve .2 g of sodium benzoate? the olubility of sodium benzoatein boiling water is 1 gram per 1.4 mL. about how much sodiumbenzoate at 25 C is 1 gram per 1.8 mL. Calculate the max mass ofsolid sodium benzoate that can be recovered when the cooledsolution is filtered.
2. why would mixed solvents be used for recrystallization?
3. explain why the melting point of pure phthalic acid is higherthan that of pure naphthalene, when both have nearly the sameweight.
Keith LeannonLv2
28 Sep 2019