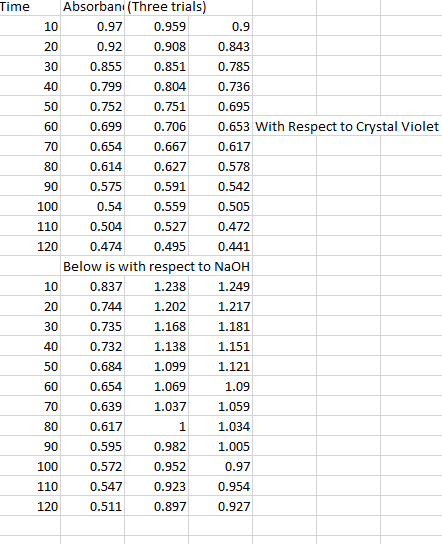1
answer
0
watching
1,103
views
28 Sep 2019
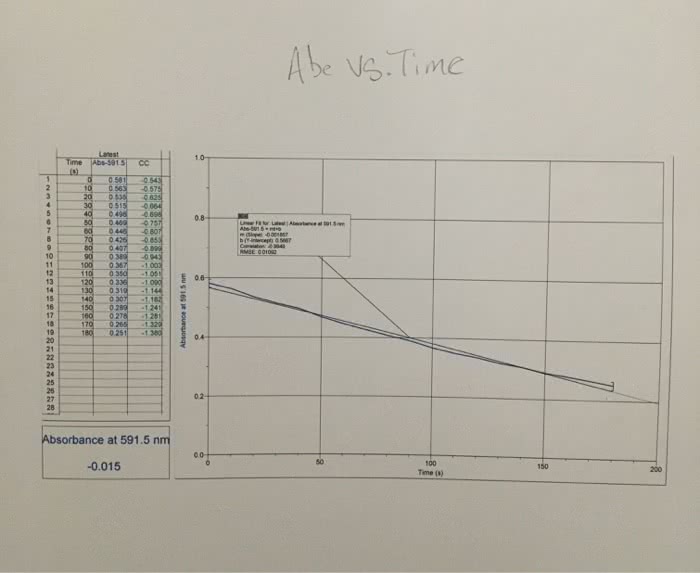
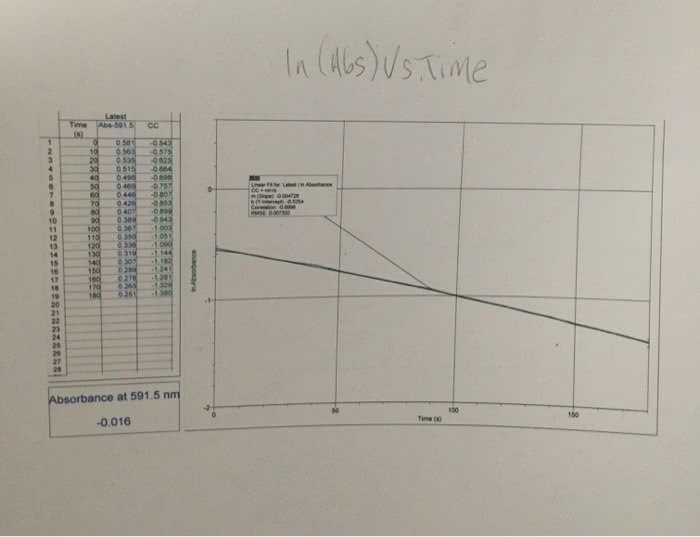
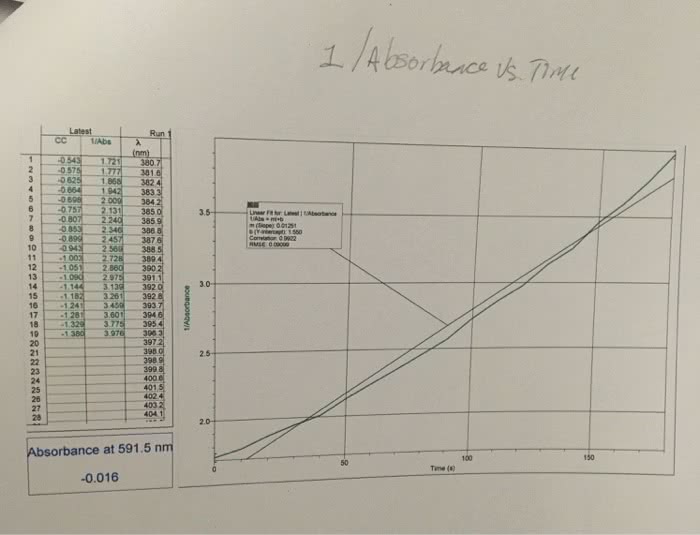
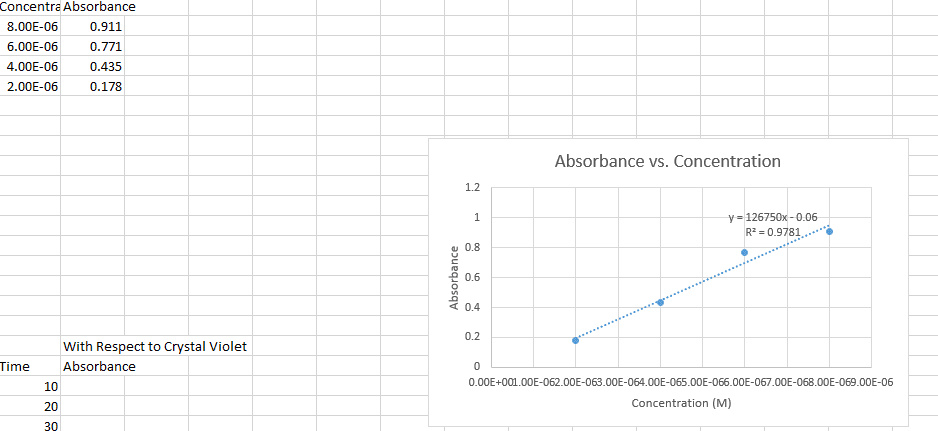
1. The rate law for a certain reaction is second order with respect to one of the reactants, R. Suppose you study this reaction, observing the absorbance of light at the analytical wavelength for R, and record the data with respect to elapsed time. Also suppose that the concentrations of all the other reactants are in large excess, and that A is the only colored species involved. Explain which absorbance function, A, ln A, or 1/A, would yield a straight-line graph when plotted against elapsed time.2. Suppose that in the reaction Q + R ? P, only the product, P, is colored.(a) What happens to the absorbance at the analytical wavelength for P of the mixture as the reaction progresses?(b) Suppose you do an experiment involving this reaction in which Q is present in large excess of R. If this reaction is first order with respect to R, would the graph of ln A (for P) versus time be a straight line? Briefly explain.Please help!
1. The rate law for a certain reaction is second order with respect to one of the reactants, R. Suppose you study this reaction, observing the absorbance of light at the analytical wavelength for R, and record the data with respect to elapsed time. Also suppose that the concentrations of all the other reactants are in large excess, and that A is the only colored species involved. Explain which absorbance function, A, ln A, or 1/A, would yield a straight-line graph when plotted against elapsed time.2. Suppose that in the reaction Q + R ? P, only the product, P, is colored.(a) What happens to the absorbance at the analytical wavelength for P of the mixture as the reaction progresses?(b) Suppose you do an experiment involving this reaction in which Q is present in large excess of R. If this reaction is first order with respect to R, would the graph of ln A (for P) versus time be a straight line? Briefly explain.Please help!
1
answer
0
watching
1,103
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nelly StrackeLv2
28 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232