2
answers
0
watching
166
views
28 Sep 2019
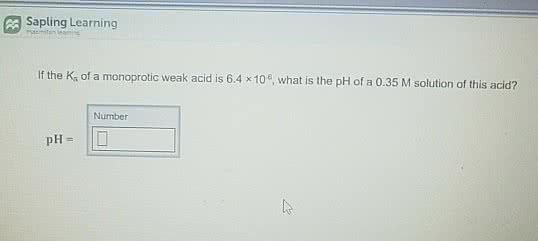
If the Ka of a monoprotic weak acid is 8.6x10^-6, what is the pH of a 0.46M solution of this acid?
If the Ka of a monoprotic weak acid is 8.6x10^-6, what is the pH of a 0.46M solution of this acid?
guruborude24Lv6
15 Apr 2023
Bunny GreenfelderLv2
28 Sep 2019
Already have an account? Log in