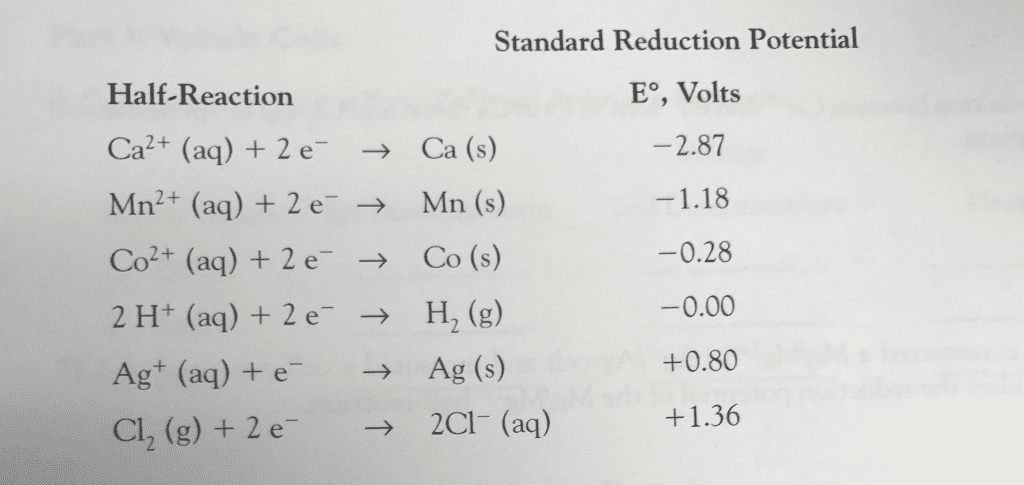1
answer
0
watching
123
views
12 Nov 2019
Given the following standard reduction potentials in acidsolution:
MnO4- + 8 H+ + 5 e- 
Co+2 + 2 e- 
Cr+3 + 3 e- 
The weakest reducing agent listed above is?
Given the following standard reduction potentials in acidsolution:
MnO4- + 8 H+ + 5 e- 
Co+2 + 2 e- 
Cr+3 + 3 e- 
The weakest reducing agent listed above is?
1
answer
0
watching
123
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Keith LeannonLv2
24 Mar 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232