(4) Rate Law from Concentration versus Time Data
NO2(g) + CO(g) --> NO(g) + CO2(g)
The rate of the above reaction depends only on the concentration of nitrogen dioxide at temperatures below 225°C. At a temperature below 225°C the following data were obtained:
Time (s)
0 1.45Ã10^3
3.62Ã10^3
5.44Ã10^3
1.09Ã10^4
2.18Ã10^4
[NO2] (mol/L)
0.580
0.554
0.519
0.493
0.428
0.340
(a) Which of the following expressions for the rate law (either differential or integrated) are completely consistent with the above experimental data. (say yes or no to each)
1/[NO2] - 1/[NO2]0 = kt
[NO2] = [NO2]0 - kt
ln[NO2] - ln[NO2]0 = -kt
-d[NO2]/dt = k[NO2]^2
k[NO2]^2 = d[CO2]/dt
(b) Calculate the rate constant (k). Enter the numerical value and corresponding units.
(4) Rate Law from Concentration versus Time Data
NO2(g) + CO(g) --> NO(g) + CO2(g)
The rate of the above reaction depends only on the concentration of nitrogen dioxide at temperatures below 225°C. At a temperature below 225°C the following data were obtained:
Time (s)
0 1.45Ã10^3
3.62Ã10^3
5.44Ã10^3
1.09Ã10^4
2.18Ã10^4
[NO2] (mol/L)
0.580
0.554
0.519
0.493
0.428
0.340
(a) Which of the following expressions for the rate law (either differential or integrated) are completely consistent with the above experimental data. (say yes or no to each)
1/[NO2] - 1/[NO2]0 = kt
[NO2] = [NO2]0 - kt
ln[NO2] - ln[NO2]0 = -kt
-d[NO2]/dt = k[NO2]^2
k[NO2]^2 = d[CO2]/dt
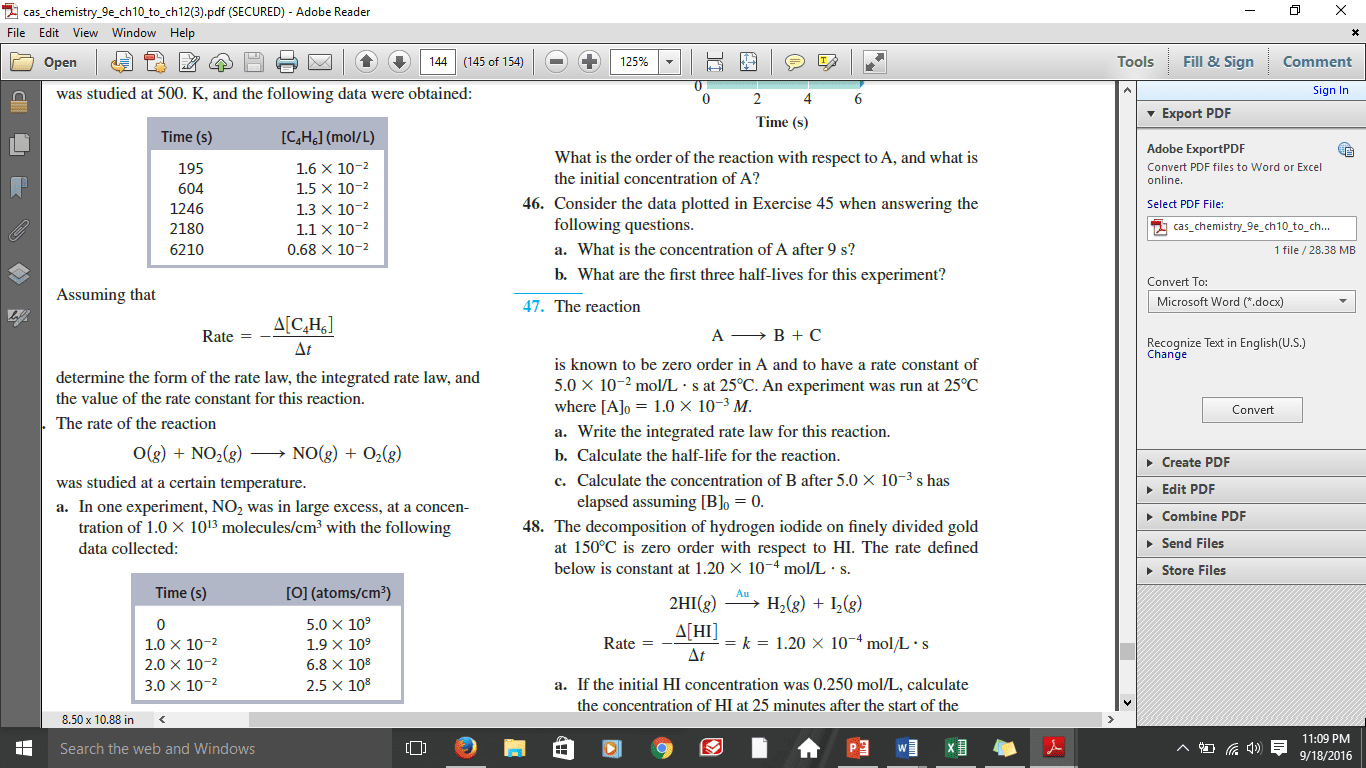
(b) Calculate the rate constant (k). Enter the numerical value and corresponding units.
For unlimited access to Homework Help, a Homework+ subscription is required.