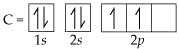
Part A) Construct an electron arrangement for Mg by indicating how many electrons are in the first, second, and third energy levels.
Enter the electron arrangement of the element by separating each energy level with a comma.
Hint 1. Example of an electron arrangement
An electron arrangement indicates the number of electrons present in each energy level of the atom. Recall that the first energy level can hold a maximum of two electrons, which the second can hold a maximum of eight electrons. The third energy level can hold a maximum of eighteen electrons, but generally, the fourth energy level will start to fill after eight electrons are in the partially filled orbital.
For example, the electron arrangement of aluminum is 2,8,3. This indicates 2 electrons in the first energy level, 8 in the second, and 3 in the third.
Part A) Construct an electron arrangement for Mg by indicating how many electrons are in the first, second, and third energy levels.
Enter the electron arrangement of the element by separating each energy level with a comma.
Hint 1. Example of an electron arrangement
An electron arrangement indicates the number of electrons present in each energy level of the atom. Recall that the first energy level can hold a maximum of two electrons, which the second can hold a maximum of eight electrons. The third energy level can hold a maximum of eighteen electrons, but generally, the fourth energy level will start to fill after eight electrons are in the partially filled orbital.
For example, the electron arrangement of aluminum is 2,8,3. This indicates 2 electrons in the first energy level, 8 in the second, and 3 in the third.