1
answer
0
watching
199
views
11 Nov 2019
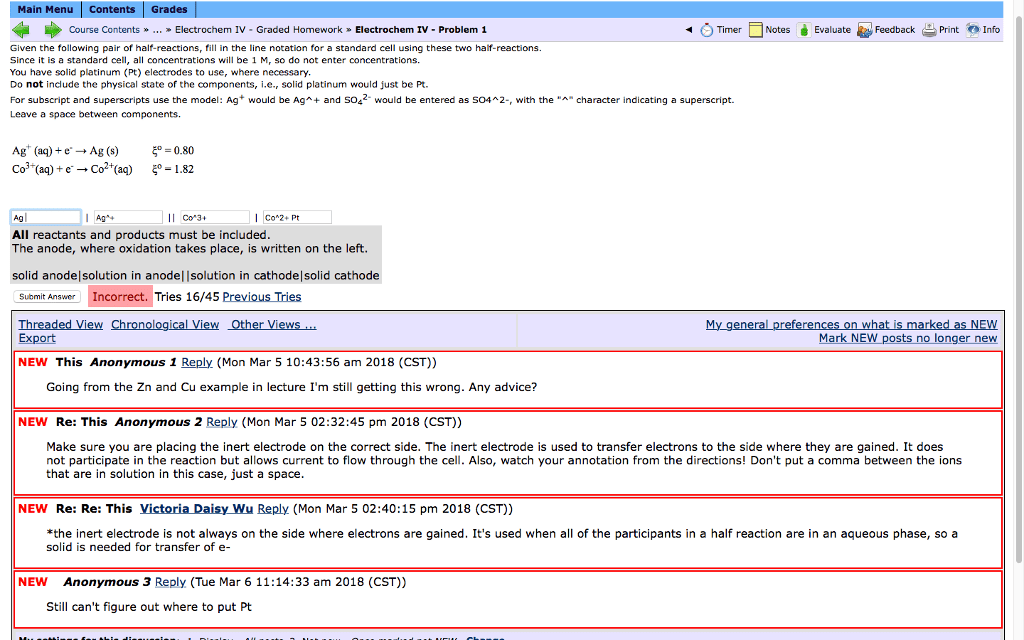
Given the following pair of half-reactions, fill in the line notation for a standard cell using these two half-reactions.
Since it is a standard cell, all concentrations will be 1 M, so do not enter concentrations.
You have solid platinum (Pt) electrodes to use, where necessary.
Do not include the physical state of the components, i.e., solid platinum would just be Pt.
For subscript and superscripts use the model: Ag+ would be Ag^+ and SO42- would be entered as SO4^2-, with the "^" character indicating a superscript.
Leave a space between components.
Tl+ (aq) + e- â Tl (s) ξo = -0.34 Au3+ (aq) + 3e- â Au (s) ξo = 1.50
Given the following pair of half-reactions, fill in the line notation for a standard cell using these two half-reactions.
Since it is a standard cell, all concentrations will be 1 M, so do not enter concentrations.
You have solid platinum (Pt) electrodes to use, where necessary.
Do not include the physical state of the components, i.e., solid platinum would just be Pt.
For subscript and superscripts use the model: Ag+ would be Ag^+ and SO42- would be entered as SO4^2-, with the "^" character indicating a superscript.
Leave a space between components.
| Tl+ (aq) + e- â Tl (s) | ξo = -0.34 | |
| Au3+ (aq) + 3e- â Au (s) | ξo = 1.50 |
Tod ThielLv2
16 Feb 2019