1
answer
0
watching
283
views
11 Nov 2019
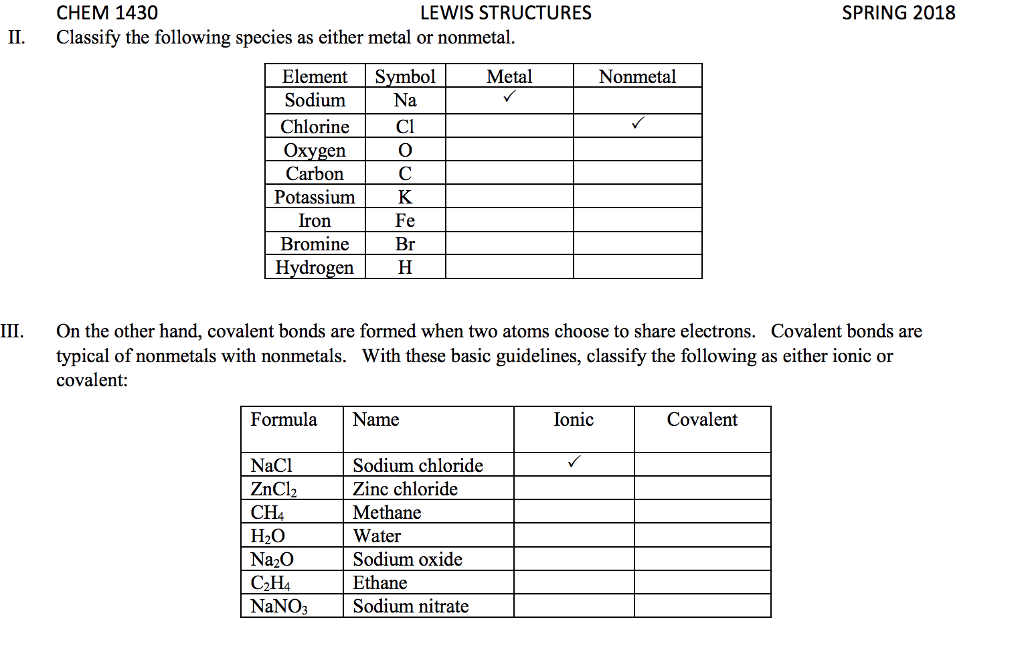
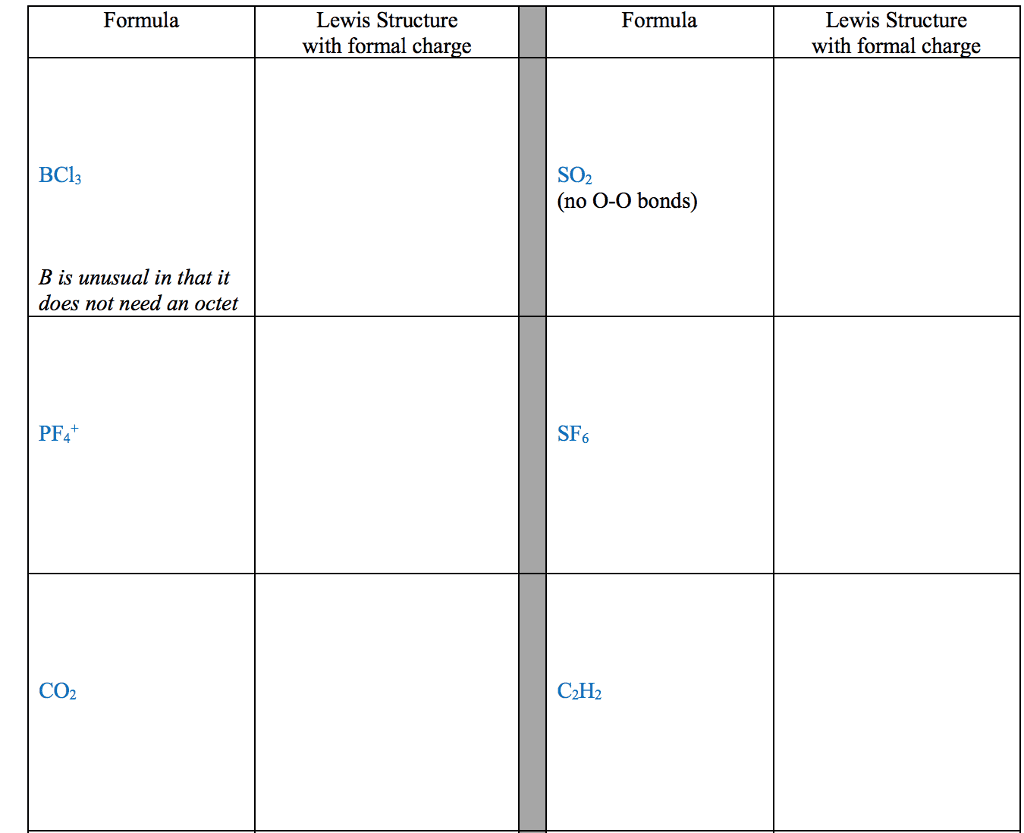
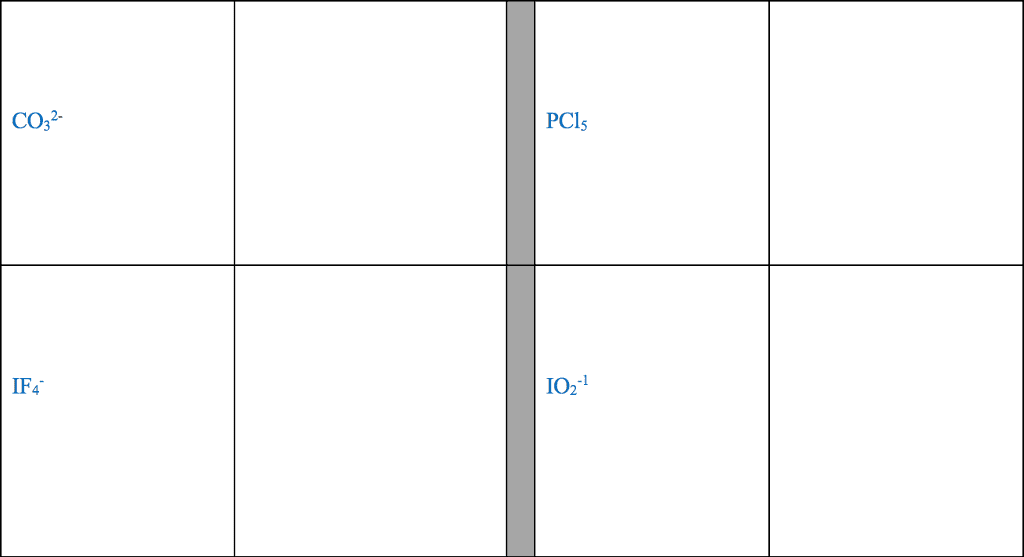
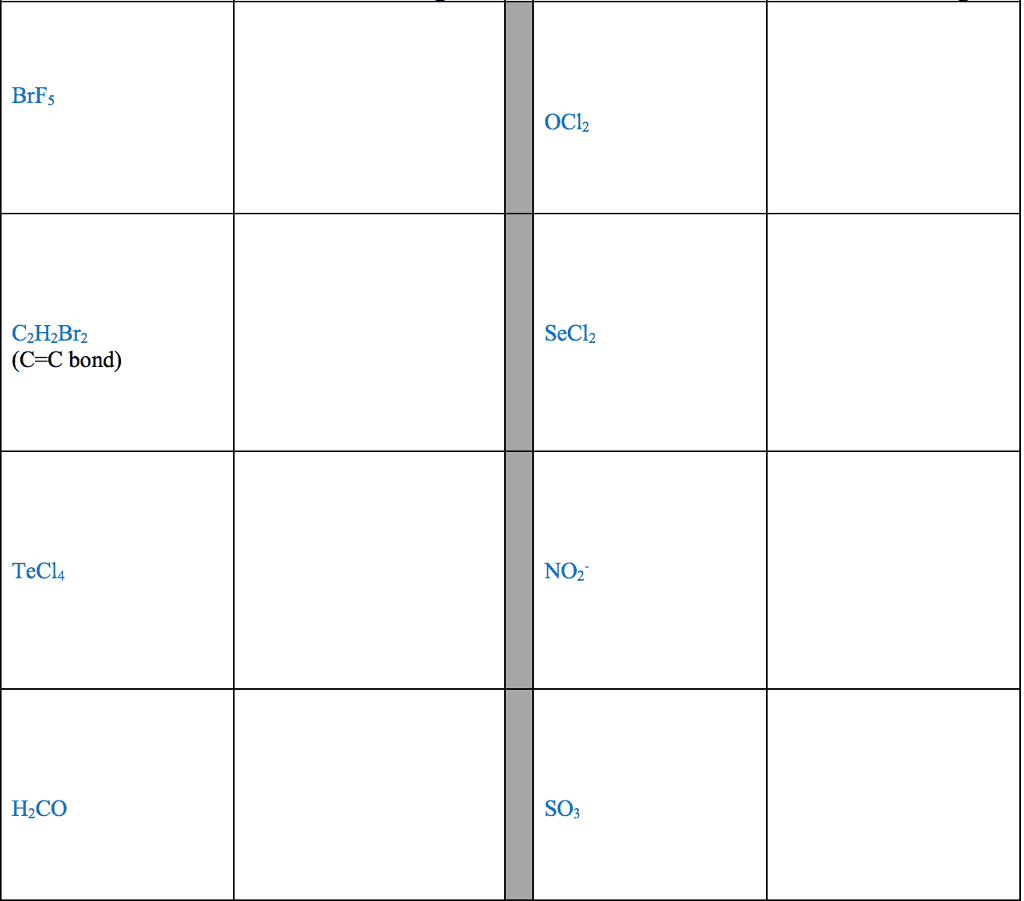
Element A has 6 valence electrons, Element B has 1 valence electron,
a) Classify each of these elements as a metal or a non- metal
b) Use Lewis symbols to show how A and B would form a compound.
c) What kind of compound is formed and what is the chemical formula for this compound
Element A has 6 valence electrons, Element B has 1 valence electron,
a) Classify each of these elements as a metal or a non- metal
b) Use Lewis symbols to show how A and B would form a compound.
c) What kind of compound is formed and what is the chemical formula for this compound
Beverley SmithLv2
31 May 2019