1
answer
0
watching
123
views
11 Nov 2019
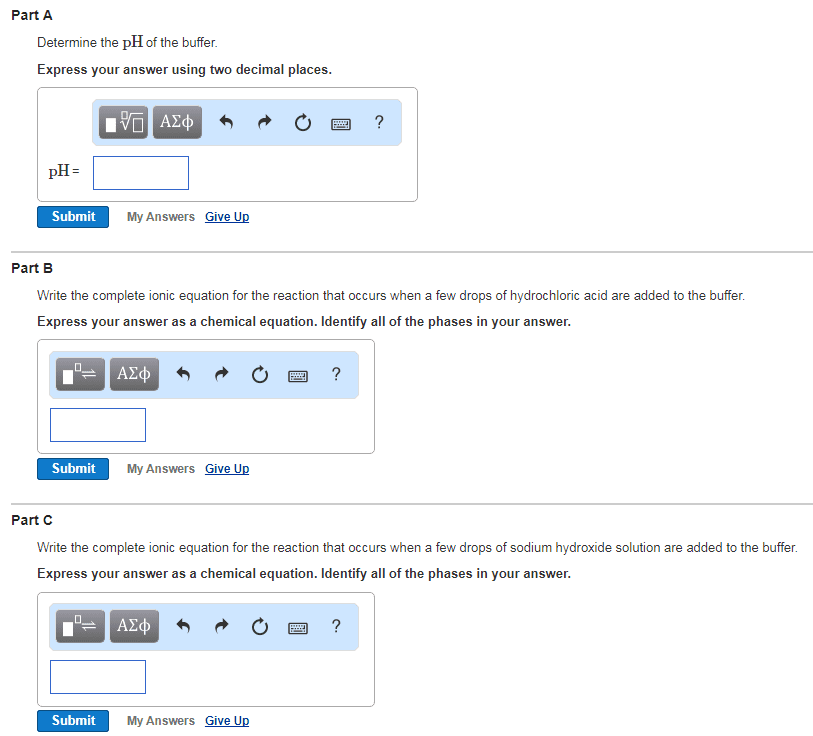
Consider a buffer solution formed by mixing 100.00mL of .1500M potassium hydrogen pthalate with 100.00mL of .0800M sodium hydroxide? As this is a buffer solution write equations to express the expected reactions.
A) Calculate the inital pH of each individual solution
B) Considering 'whats in solution' write the chemistry that occurs when the solutions are mixed (two lines of chemistry are required)
Consider a buffer solution formed by mixing 100.00mL of .1500M potassium hydrogen pthalate with 100.00mL of .0800M sodium hydroxide? As this is a buffer solution write equations to express the expected reactions.
A) Calculate the inital pH of each individual solution
B) Considering 'whats in solution' write the chemistry that occurs when the solutions are mixed (two lines of chemistry are required)
Patrina SchowalterLv2
8 Apr 2019