2
answers
0
watching
280
views
28 Sep 2019
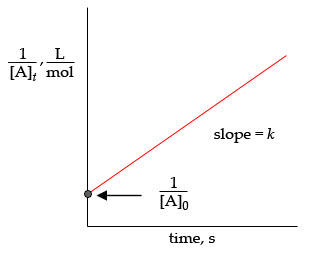
Consider the second-order reaction: 2NO2(g) ---> 2NO (g) +O2(g)
Find the initial concentration [NO2]0 and the rate constant k forthe reaction. What will be the concentration of NO2 after t = 30.0s([NO2]t) for a reaction with this information:
Rate law: k[NO2]2
k=.541/(mol/s) at 573k
initial rate= 1.35 mol/(ls)
rate=0.13 mol/(ls)
[NO2]= 0.49 mol/l
Consider the second-order reaction: 2NO2(g) ---> 2NO (g) +O2(g)
Find the initial concentration [NO2]0 and the rate constant k forthe reaction. What will be the concentration of NO2 after t = 30.0s([NO2]t) for a reaction with this information:
Rate law: k[NO2]2
k=.541/(mol/s) at 573k
initial rate= 1.35 mol/(ls)
rate=0.13 mol/(ls)
[NO2]= 0.49 mol/l
Find the initial concentration [NO2]0 and the rate constant k forthe reaction. What will be the concentration of NO2 after t = 30.0s([NO2]t) for a reaction with this information:
Rate law: k[NO2]2
k=.541/(mol/s) at 573k
initial rate= 1.35 mol/(ls)
rate=0.13 mol/(ls)
[NO2]= 0.49 mol/l
25 Apr 2023
Hubert KochLv2
28 Sep 2019
Already have an account? Log in