1
answer
0
watching
418
views
11 Nov 2019
1) Assuming ideal behavior, what is the ionic concentration of a 0.135 M solution of trisodium phosphate?
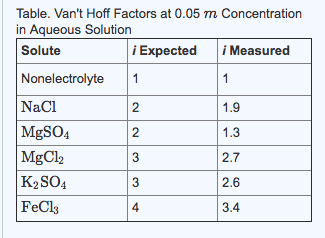
2) What is the boiling point of a solution (in oC) that was prepared by dissolving 13.38 g of MgCl2 in 250.0 g water. MgCl2 is an ionic solid with a measured van't Hoff factor of 2.7. KbH2O= 0.52 oC.kg/mol
1) Assuming ideal behavior, what is the ionic concentration of a 0.135 M solution of trisodium phosphate?
2) What is the boiling point of a solution (in oC) that was prepared by dissolving 13.38 g of MgCl2 in 250.0 g water. MgCl2 is an ionic solid with a measured van't Hoff factor of 2.7. KbH2O= 0.52 oC.kg/mol
Hubert KochLv2
20 Feb 2019