1
answer
0
watching
1,479
views
28 Sep 2019
1) Hydrates contain water in a stoichiometric quantity. Whatdoes this mean?
2) Why can you use calcium chloride to reduce the moisture in ahumid basement area?
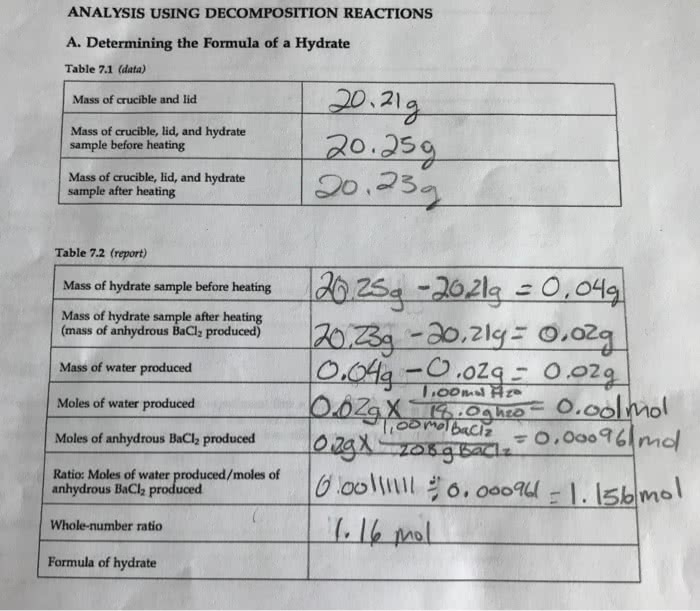
3) Plaster of Paris is calcium sulfate monohydrate,(CaSO4)2.H2O. (A) How many total atoms of all kinds are present?(B) On heating the hydrate, how many moles of water should bedriven off per mole of hydrate? (C) Calculate the percent of waterin the hydrate. SHOW YOUR WORK. (D) If you heat 20.00g of hydrateand drive off the water, what is the mass of the anhydrous saltremaining? SHOW YOUR WORK.
1) Hydrates contain water in a stoichiometric quantity. Whatdoes this mean?
2) Why can you use calcium chloride to reduce the moisture in ahumid basement area?
3) Plaster of Paris is calcium sulfate monohydrate,(CaSO4)2.H2O. (A) How many total atoms of all kinds are present?(B) On heating the hydrate, how many moles of water should bedriven off per mole of hydrate? (C) Calculate the percent of waterin the hydrate. SHOW YOUR WORK. (D) If you heat 20.00g of hydrateand drive off the water, what is the mass of the anhydrous saltremaining? SHOW YOUR WORK.
Liked by kloiebright
Nestor RutherfordLv2
28 Sep 2019