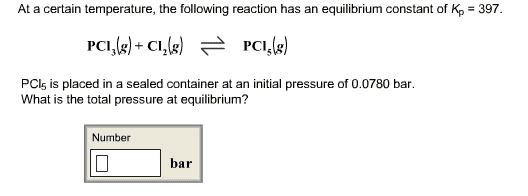1
answer
0
watching
197
views
10 Nov 2019
at a certain temperature the following reaction has anequilibruim constant of kp=397
PCI3(g) + CI2(g) <--->PCI5(g)
CI5 is placed in a sealed container at an initial pressure of0.0680 bar. what is the total pressure of the equilibrium
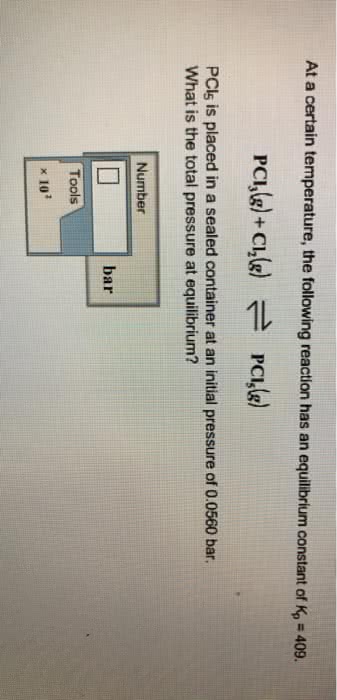
at a certain temperature the following reaction has anequilibruim constant of kp=397
PCI3(g) + CI2(g) <--->PCI5(g)
CI5 is placed in a sealed container at an initial pressure of0.0680 bar. what is the total pressure of the equilibrium
Tod ThielLv2
10 Nov 2019