1
answer
0
watching
77
views
10 Nov 2019
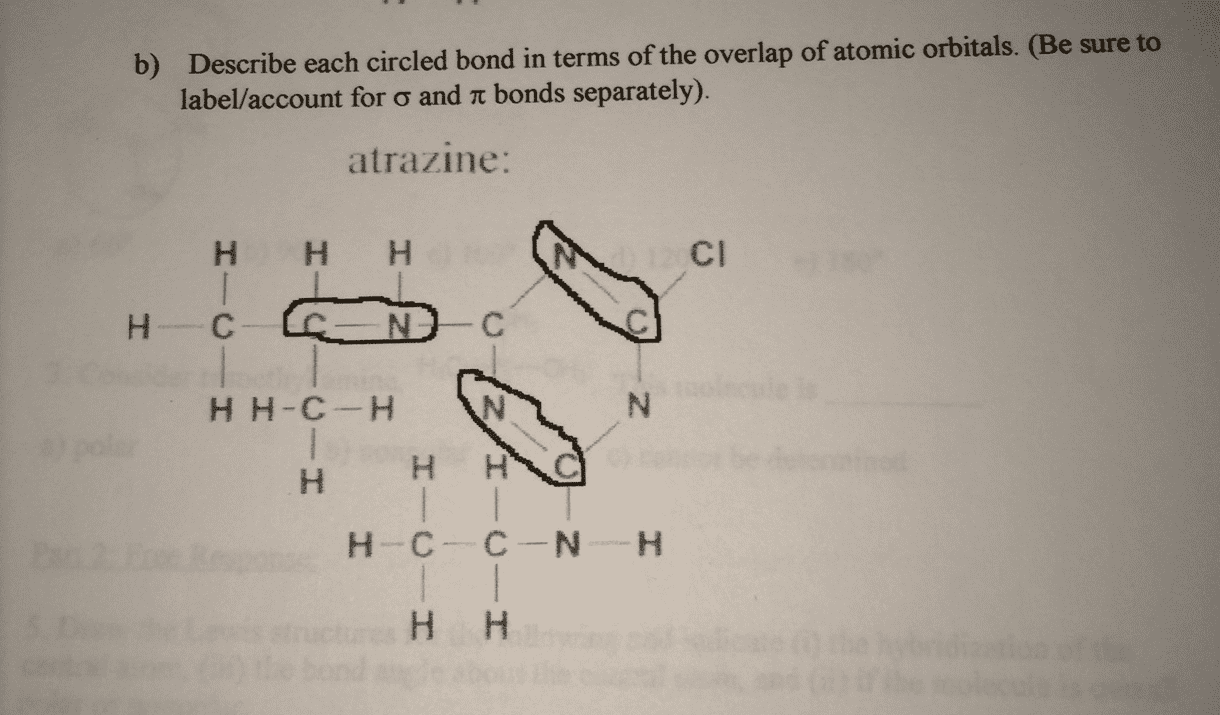
Describe each circled bond in terms of the overlap of atomic orbitals. (Be sure to label/account for sigma and pi bonds separately).

Describe each circled bond in terms of the overlap of atomic orbitals. (Be sure to label/account for sigma and pi bonds separately).
Reid WolffLv2
3 Nov 2019
