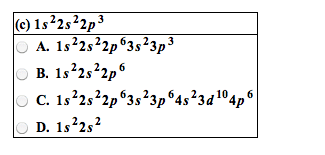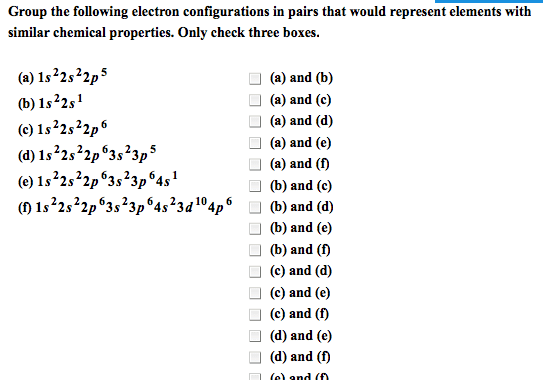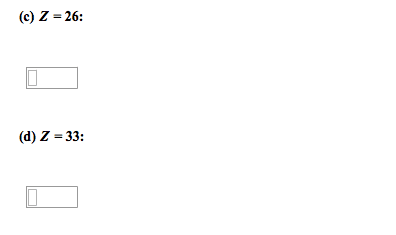2
answers
1
watching
2,219
views
10 Nov 2019
Sodium and potassium have similar chemical and physical properties. This is best explained by the fact that both elements
have the same ground-state valence-electron configuration.
have low relative atomic masses.
are in Period 1 of the periodic table.
have relatively low first ionization energies.
are active metals.
Sodium and potassium have similar chemical and physical properties. This is best explained by the fact that both elements
| have the same ground-state valence-electron configuration. | ||
| have low relative atomic masses. | ||
| are in Period 1 of the periodic table. | ||
| have relatively low first ionization energies. | ||
| are active metals. |
2
answers
1
watching
2,219
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Read by 12 people
27 Sep 2021
Bunny GreenfelderLv2
22 Apr 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232