1
answer
0
watching
764
views
10 Nov 2019
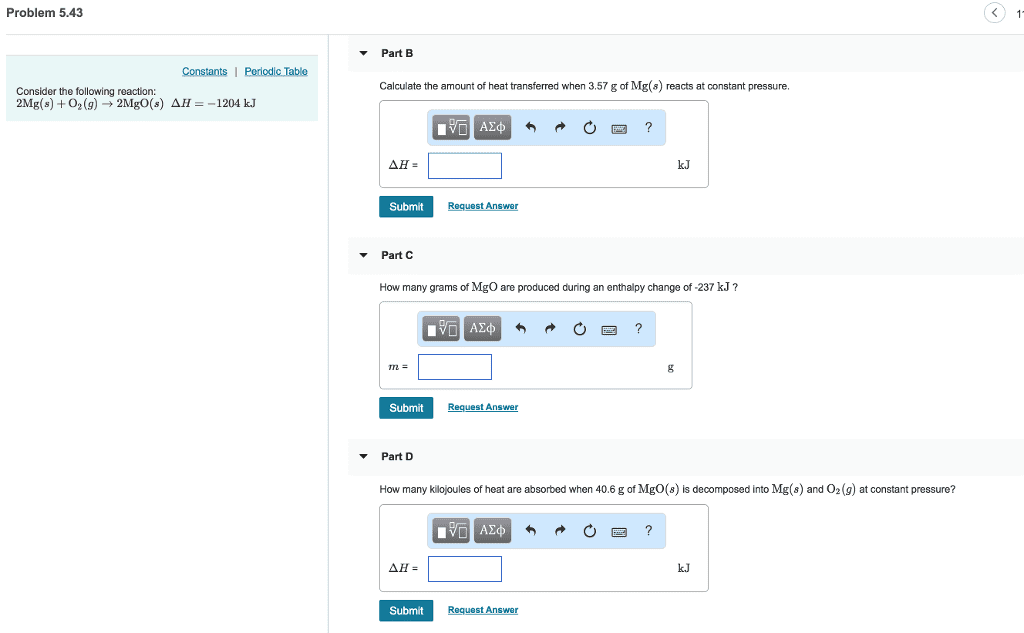
2 Mg(s) + O2(g) ? 2 MgO(s) ?H = ?1204 kJ(a) Is this reaction exothermic or endothermic?exothermicendothermic (b) Calculate the amount of heat transferred when 2.1 g of Mg(s) reacts at constant pressure. kJ (c) How many grams of MgO are produced during an enthalpy change of 96.0 kJ? g (d) How many kilojoules of heat are absorbed when 7.30 g of MgO(s) are decomposed into Mg(s) and O2(g) at constant pressure? kJ
2 Mg(s) + O2(g) ? 2 MgO(s) ?H = ?1204 kJ(a) Is this reaction exothermic or endothermic?exothermicendothermic (b) Calculate the amount of heat transferred when 2.1 g of Mg(s) reacts at constant pressure. kJ (c) How many grams of MgO are produced during an enthalpy change of 96.0 kJ? g (d) How many kilojoules of heat are absorbed when 7.30 g of MgO(s) are decomposed into Mg(s) and O2(g) at constant pressure? kJ
Collen VonLv2
11 Apr 2019