1
answer
0
watching
127
views
6 Nov 2019
Balancethe following equations. (Use the lowest possible whole numbercoefficients. Include states-of-matter under the given conditionsin your answer.)
(a) KNO3(s)?K2O(s) + N2O5(g)
(b) C10H22(l) + O2(g) ?CO2(g) + H2O(g)
(c) Fe(s)+ S8(s) ?FeS(s)
(d) RbClO3(s)?RbCl(s) + O2(g)
(e) Eu2O3(s)+ H2S(g) ?EuS(s) + H2O(g) +S(g)
Balancethe following equations. (Use the lowest possible whole numbercoefficients. Include states-of-matter under the given conditionsin your answer.)
(a) KNO3(s)?K2O(s) + N2O5(g)
(b) C10H22(l) + O2(g) ?CO2(g) + H2O(g)
(c) Fe(s)+ S8(s) ?FeS(s)
(d) RbClO3(s)?RbCl(s) + O2(g)
(e) Eu2O3(s)+ H2S(g) ?EuS(s) + H2O(g) +S(g)
1
answer
0
watching
127
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Elin HesselLv2
19 Feb 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
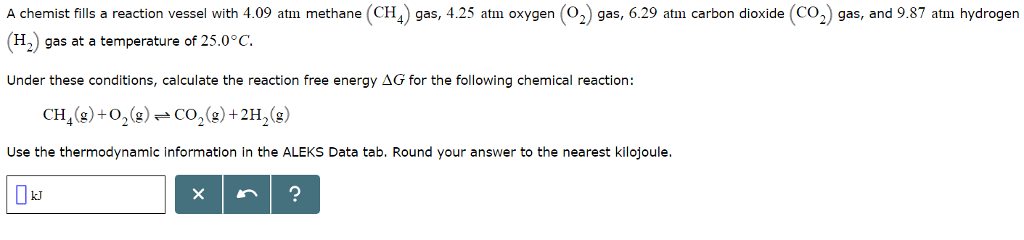
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
A chemist fills a reaction vessel with 4.09 atm methane (CH gas, 4.25 atm oxygen (2) gas, 6.29 atm carbon dioxide (CO2 gas, and 9.87 atm hydrogen (H2) gas at a temperature of 25.0° C. Under these conditions, calculate the reaction free energy ÎG for the following chemical reaction: CH4(g) +02(g)--CO2(g)+2H2(g) Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule
