1
answer
0
watching
239
views
6 Nov 2019
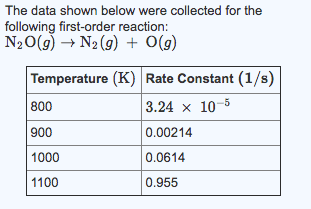
#21) The data shown below were collected for the followingsecond-order reaction:
Cl(g)+H2(g)->HCl(g)+H(g)
Temperature (K) Rate Constant (L/mol*s
90 0.00357
100 0.0773
110 0.956
120 7.781
1) Use an Arrhenius plot to determine the activation barrier forthe reaction.
Ea=_________Kj/mol
2) Use an Arrhenius plot to determine the frequency factor for thereaction.
A=_________s^-1
#21) The data shown below were collected for the followingsecond-order reaction:
Cl(g)+H2(g)->HCl(g)+H(g)
Temperature (K) Rate Constant (L/mol*s
90 0.00357
100 0.0773
110 0.956
120 7.781
1) Use an Arrhenius plot to determine the activation barrier forthe reaction.
Ea=_________Kj/mol
2) Use an Arrhenius plot to determine the frequency factor for thereaction.
A=_________s^-1
Cl(g)+H2(g)->HCl(g)+H(g)
Temperature (K) Rate Constant (L/mol*s
90 0.00357
100 0.0773
110 0.956
120 7.781
1) Use an Arrhenius plot to determine the activation barrier forthe reaction.
Ea=_________Kj/mol
2) Use an Arrhenius plot to determine the frequency factor for thereaction.
A=_________s^-1
Patrina SchowalterLv2
1 Jun 2019