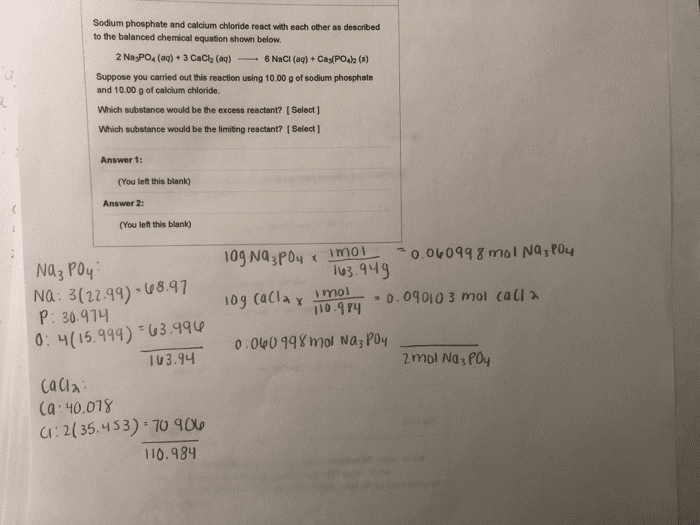2
answers
2
watching
2,164
views
limefrog525Lv1
6 Nov 2019
Phosphoric acid can be prepared by reaction of sulfuric acid with"phosphate rock" according to the equation: Ca3(PO4)2+3H2SO4---> 3CaSO4 + 2H3PO4
What is the molar mass of Ca3(PO4)2?
How many oxygen atoms are there in 1.55mg of Ca3(PO4)2?
Suppose the reaction is carried out starting with 103 g ofCa3(PO4)2 and 75.0 g of H2SO4. Which substance is the limitingreactant?
Phosphoric acid can be prepared by reaction of sulfuric acid with"phosphate rock" according to the equation: Ca3(PO4)2+3H2SO4---> 3CaSO4 + 2H3PO4
What is the molar mass of Ca3(PO4)2?
How many oxygen atoms are there in 1.55mg of Ca3(PO4)2?
Suppose the reaction is carried out starting with 103 g ofCa3(PO4)2 and 75.0 g of H2SO4. Which substance is the limitingreactant?
What is the molar mass of Ca3(PO4)2?
How many oxygen atoms are there in 1.55mg of Ca3(PO4)2?
Suppose the reaction is carried out starting with 103 g ofCa3(PO4)2 and 75.0 g of H2SO4. Which substance is the limitingreactant?
2
answers
2
watching
2,164
views
For unlimited access to Homework Help, a Homework+ subscription is required.
26 Apr 2023
Jean KeelingLv2
15 Oct 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232