4
answers
1
watching
2,130
views
26 Oct 2017
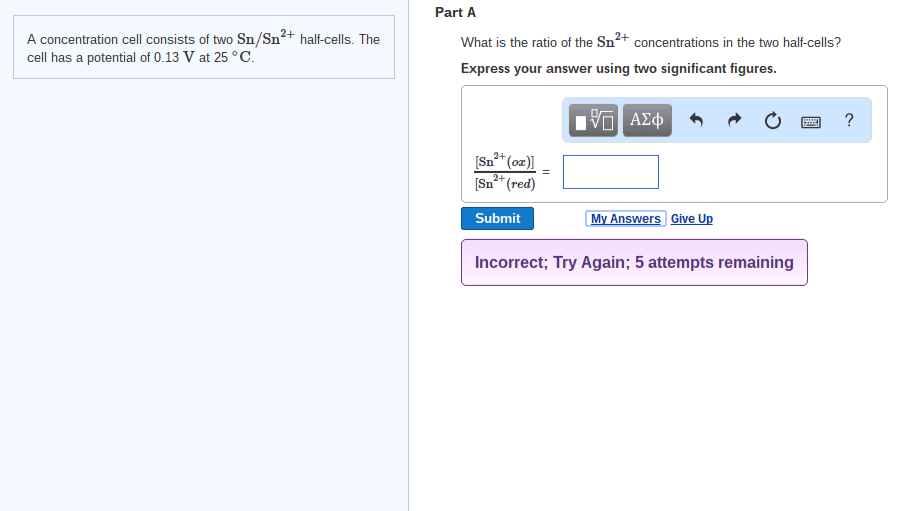
81. A concentration cell consists of two Sn/Sn2+ half-cells. The cell has a potential of 0.10 V at 25°C. What is the ratio of the Sn2+ concentrations in the two half-cells?
81. A concentration cell consists of two Sn/Sn2+ half-cells. The cell has a potential of 0.10 V at 25°C. What is the ratio of the Sn2+ concentrations in the two half-cells?
jagdishkumarLv7
15 Jul 2023
Already have an account? Log in
Reid WolffLv2
26 Oct 2017
Already have an account? Log in