1
answer
0
watching
123
views
6 Nov 2019
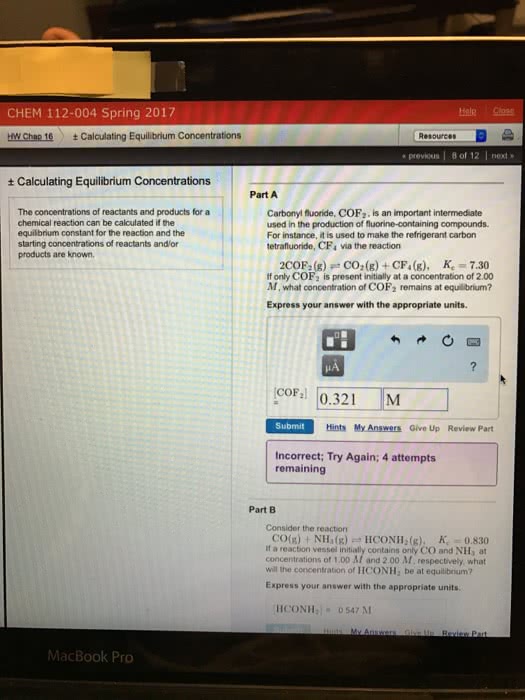
arbonyl fluoride, \rm COF_2, is an important intermediate used inthe production of fluorine-containing compounds. For instance, itis used to make the refrigerant carbon tetrafluoride, \rm CF_4 viathe reaction
\rm 2 COF_2(g) \rightleftharpoons CO_2(g) + CF_4(g),~~~~{\itK}_c=6.70
If only \rm COF_2 is present initially at a concentration of 2.00M, what concentration of \rm COF_2 remains at equilibrium?
Express your answer with the appropriate units.
\rm [COF_2] =
Try Again; 7 attempts remaining
submithintsmy answers
Part B
Consider the reaction
\rm CO(g) + NH_3(g) \rightleftharpoons HCONH_2(g),~~~~{\itK}_c=0.900
If a reaction vessel initially contains only \rm CO and \rm NH_3 atconcentrations of 1.00 M and 2.00 M, respectively, what will theconcentration of \rm HCONH_2 be at equilibrium?
Express your answer with the appropriate units.
\rm [HCONH_2] =
submithints
arbonyl fluoride, \rm COF_2, is an important intermediate used inthe production of fluorine-containing compounds. For instance, itis used to make the refrigerant carbon tetrafluoride, \rm CF_4 viathe reaction
\rm 2 COF_2(g) \rightleftharpoons CO_2(g) + CF_4(g),~~~~{\itK}_c=6.70
If only \rm COF_2 is present initially at a concentration of 2.00M, what concentration of \rm COF_2 remains at equilibrium?
Express your answer with the appropriate units.
\rm [COF_2] =
Try Again; 7 attempts remaining
submithintsmy answers
Part B
Consider the reaction
\rm CO(g) + NH_3(g) \rightleftharpoons HCONH_2(g),~~~~{\itK}_c=0.900
If a reaction vessel initially contains only \rm CO and \rm NH_3 atconcentrations of 1.00 M and 2.00 M, respectively, what will theconcentration of \rm HCONH_2 be at equilibrium?
Express your answer with the appropriate units.
\rm [HCONH_2] =
submithints
\rm 2 COF_2(g) \rightleftharpoons CO_2(g) + CF_4(g),~~~~{\itK}_c=6.70
If only \rm COF_2 is present initially at a concentration of 2.00M, what concentration of \rm COF_2 remains at equilibrium?
Express your answer with the appropriate units.
\rm [COF_2] =
Try Again; 7 attempts remaining
submithintsmy answers
Part B
Consider the reaction
\rm CO(g) + NH_3(g) \rightleftharpoons HCONH_2(g),~~~~{\itK}_c=0.900
If a reaction vessel initially contains only \rm CO and \rm NH_3 atconcentrations of 1.00 M and 2.00 M, respectively, what will theconcentration of \rm HCONH_2 be at equilibrium?
Express your answer with the appropriate units.
\rm [HCONH_2] =
submithints
Beverley SmithLv2
23 Oct 2019