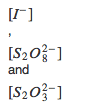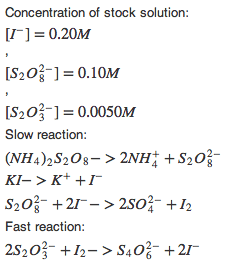2
answers
0
watching
134
views
rubyyak737Lv1
6 Nov 2019
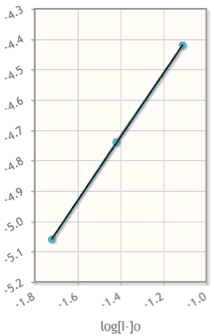
So I need to find the concentration of I2(aq)
The reactions are;
2I-(aq)+S2O82--->2SO42-+I2(aq)
2S2O32-(aq)+I2(aq)-->S4O62-(aq)+2I-(aq)
The concentration for KI is 0.0401 mol/L, for(NH4)2S2O8 is 0.0602mol/L and for Na2S2O3 is4.17x10-4mol/L.
4.04 mL of KI, 4.04 mL of(NH4)2S2O8, 2.01 mL ofNa2S2O3 are used
I know I should be using the second equation, I find the molesfor I2 but I cannot figure the concentration
So I need to find the concentration of I2(aq)
The reactions are;
2I-(aq)+S2O82--->2SO42-+I2(aq)
2S2O32-(aq)+I2(aq)-->S4O62-(aq)+2I-(aq)
The concentration for KI is 0.0401 mol/L, for(NH4)2S2O8 is 0.0602mol/L and for Na2S2O3 is4.17x10-4mol/L.
4.04 mL of KI, 4.04 mL of(NH4)2S2O8, 2.01 mL ofNa2S2O3 are used
I know I should be using the second equation, I find the molesfor I2 but I cannot figure the concentration
2
answers
0
watching
134
views
For unlimited access to Homework Help, a Homework+ subscription is required.
oldsheldonLv5
2 May 2024
Jean KeelingLv2
30 Jun 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232