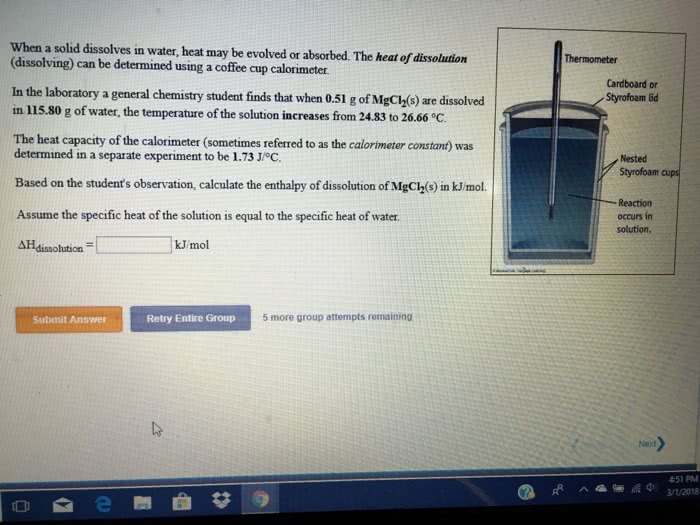
1
answer
0
watching
176
views
28 Sep 2019
0.325 mol of a solid was dissolved in 300 mL of water at 25.5oC in a styrofoam cup calorimeter. After the solid hadfully dissolved, the final temperature of the solution was 22.3oC. What is the molar heat of solution of thesubstance?
Report your answer to the nearest 0.1 kJ.
1 kJ/mol
What is the specific heat of a metal if addition of 70.0 g of themetal at 17.0 oC to 210.0 g of Cu (s = 0.38J/g-oC) at 193.5 oC produces a mixture thatreaches thermal equilibrium at 162.0 oC? s =1 J/g·oC
0.325 mol of a solid was dissolved in 300 mL of water at 25.5oC in a styrofoam cup calorimeter. After the solid hadfully dissolved, the final temperature of the solution was 22.3oC. What is the molar heat of solution of thesubstance?
Report your answer to the nearest 0.1 kJ.
What is the specific heat of a metal if addition of 70.0 g of themetal at 17.0 oC to 210.0 g of Cu (s = 0.38J/g-oC) at 193.5 oC produces a mixture thatreaches thermal equilibrium at 162.0 oC? s =1 J/g·oC
Beverley SmithLv2
28 Sep 2019