4
answers
0
watching
1,295
views
19 Dec 2017
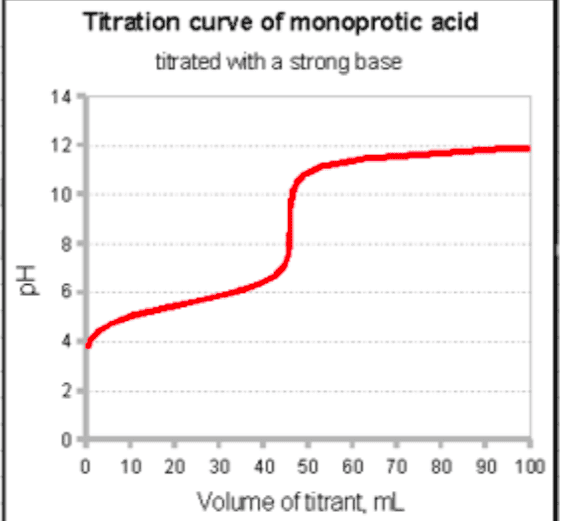
78. A 0.446-g sample of an unknown monoprotic acid is titrated with 0.105 M KOH. The resulting titration curve is shown here. Determine the molar mass and pK, of the acid. pH OOO 0 20 40 60 80 100 Volume of base added (mL)
78. A 0.446-g sample of an unknown monoprotic acid is titrated with 0.105 M KOH. The resulting titration curve is shown here. Determine the molar mass and pK, of the acid. pH OOO 0 20 40 60 80 100 Volume of base added (mL)
jagdishkumarLv7
15 Jul 2023
16 Jun 2023
Already have an account? Log in
Trinidad TremblayLv2
21 Dec 2017
Already have an account? Log in