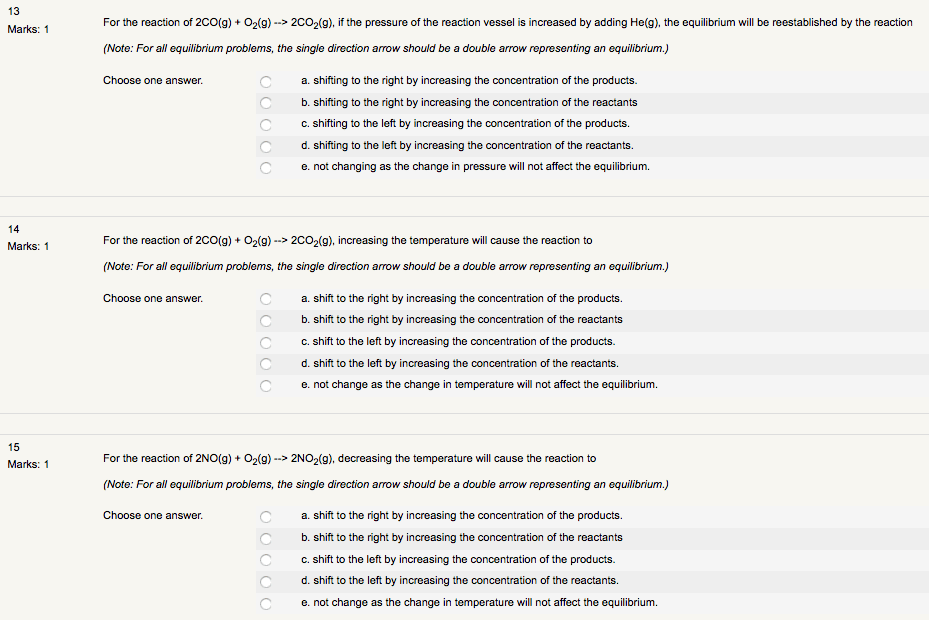
Consider the following equilibrium for the next fivequestions:
2NO (g) + O2 (g) <--> 2NO2(g) ÎH< 0
Use the following choices and place correct letter next toproper description:
A. Shift Left B. Shift Right C. NoChange D. NotEnough Information
______ 1 mole of NO2(g) is added
______ 1 mole of NO (g) isremoved
______ 1 mole of He (g) isadded
______ The pressure is doubled byhalving the volume.
______ The temperature isincreased
Consider the following equilibrium for the next fivequestions:
2NO (g) + O2 (g) <--> 2NO2(g) ÎH< 0
Use the following choices and place correct letter next toproper description:
A. Shift Left B. Shift Right C. NoChange D. NotEnough Information
______ 1 mole of NO2(g) is added
______ 1 mole of NO (g) isremoved
______ 1 mole of He (g) isadded
______ The pressure is doubled byhalving the volume.
______ The temperature isincreased
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Consider the following equilibrium, for which ?H< 0
2SO2(g)+O2(g)?2SO3(g)
How will each of the following changes affect an equilibrium mixture of the three gases
Part C
the volume of the reaction vessel is doubled
| A) shift equilibrium to the right | |
| B) shift equilibrium to the left | |
| C) no affect |
Part D
a catalyst is added to the mixture
| A) | shift equilibrium to the right |
| B) | shift equilibrium to the left |
| C) | no affect |
Part E
the total pressure of the system is increased by adding a noble gas
| A) | shift equilibrium to the right |
| B) | shift equilibrium to the left |
| C) | no affect |
Part F
SO3(g) is removed from the system
| A) | shift equilibrium to the right |
| B) | shift equilibrium to the left |
| C) | no affect |