2
answers
0
watching
689
views
28 Sep 2019
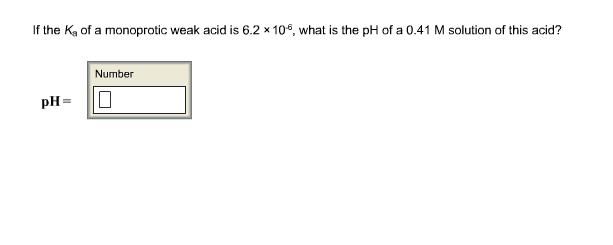
If the Ka of a monoprotic weak acid is 4.9 Ã 10-6, what is the pH of a 0.50 M solution of this acid? Can you write out the answer and take a picture? Thanks.
If the Ka of a monoprotic weak acid is 4.9 Ã 10-6, what is the pH of a 0.50 M solution of this acid? Can you write out the answer and take a picture? Thanks.
guruborude24Lv6
15 Apr 2023
Hubert KochLv2
30 Sep 2019
Already have an account? Log in