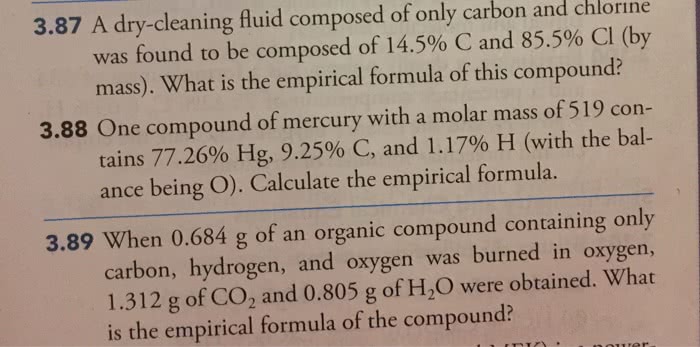1
answer
0
watching
163
views
olivebee559Lv1
28 Aug 2019
- Use the information provided to determine the empirical and molecular formula for each of the following:
a. 39% carbon, 3.47% hydrogen, and 55.14% oxygen; experimental molar mass: 116.07g
b. 53% carbon, 9.15% hydrogen, and 36.32% oxygen; experimental molar mass: 88g;
c. 27% carbon, 7.19% hydrogen, and 28.54% oxygen; experimental molar mass 168.19g
d. A hydrocarbon containing 17.4% hydrogen by mass, with a molar mass of 58.1g.
- Use the information provided to determine the empirical and molecular formula for each of the following:
a. 39% carbon, 3.47% hydrogen, and 55.14% oxygen; experimental molar mass: 116.07g
b. 53% carbon, 9.15% hydrogen, and 36.32% oxygen; experimental molar mass: 88g;
c. 27% carbon, 7.19% hydrogen, and 28.54% oxygen; experimental molar mass 168.19g
d. A hydrocarbon containing 17.4% hydrogen by mass, with a molar mass of 58.1g.
Read by 1 person
2 Jun 2021