CHEM 201 Lecture Notes - Lecture 23: Freezing-Point Depression, Colligative Properties, Molality
12 views2 pages
Document Summary
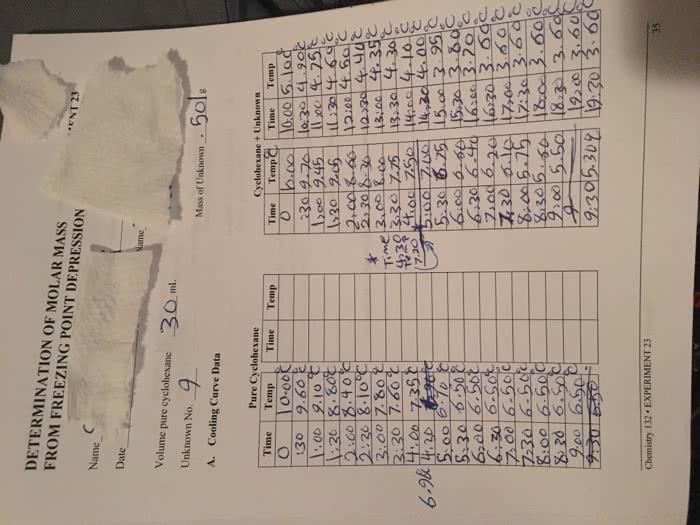
Colligative properties are solution properties which depend on the collective concentration of all solutes: colligative properties are about the solvent, not the solute. Elevation of boiling point: for a solution, there is less evp to start with, so a higher temperature is needed for boiling the solvent in the solution. Tf=fp of pure solvent -fp of actual solution. *used to calculate how much the freezing point went down. Kf=freezing point depression constant with units of c/m or k/m m=molality concentration of the solute. *used to calculate how much the freezing point went down: tf can be used to calculate the molar mass of an unknown. Steps: use the previous 2 equations to find molality, times molality by the mass (in kg) of the molecule that you dissolve the molecule into, divide the gram of solute by this number. =m*r*t: molar mass can be calculated from osmotic pressure:
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232